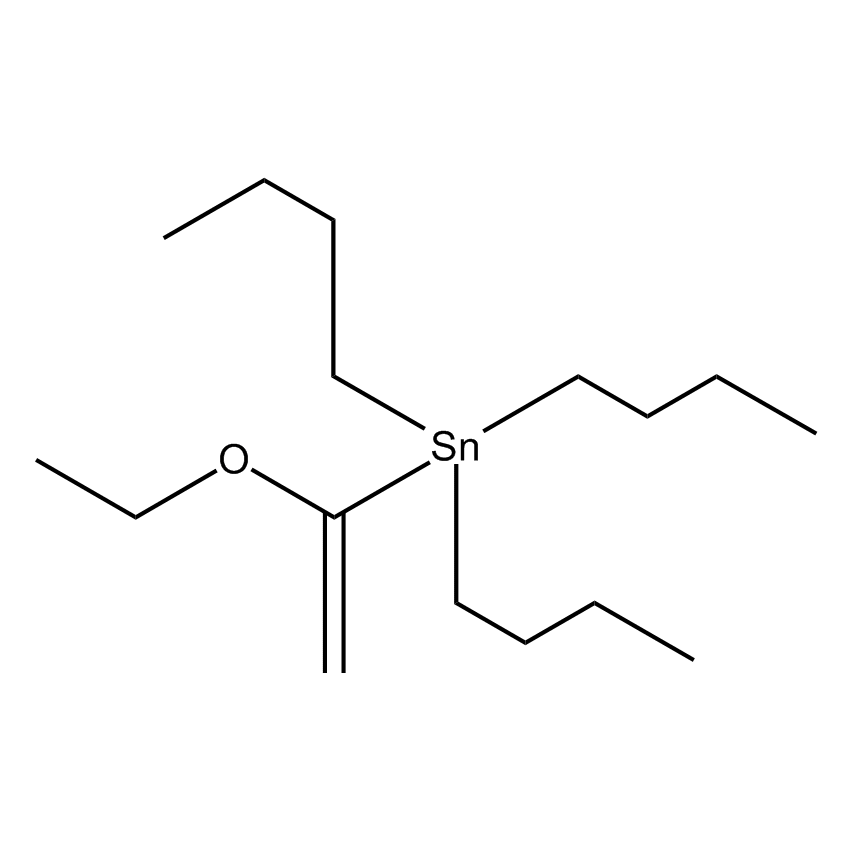
Can be cross-coupled with aryl chlorides.1
Used to acylate aryl triflates.2
Undergoes Stille cross-coupling reactions as employed in the syntheses of three cladiellins.3
Cross-couples with vinyl sulfides.4
Can be cross-coupled with heteroaromatic chlorides.5
1. Soderquist, J. et al. Tetrahedron Lett. 1983, 24, 2361.
2. Stoltz, B. M. et al. J. Am. Chem. Soc. 2000, 122, 9044.
3. Clark, J. S. et al. Angew. Chem., Int. Ed. Engl. 2010, 49, 9867.
4. Schleiss, J.; Rollin, P.; Tatibouet, P. Angew. Chem., Int. Ed. Engl. 2010, 49, 577.
5. Hong. L. et al. Bioorg. Med. Chem. Lett. 2010, 20, 679.
Organotin Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
1-Ethoxyvinyltri-n-butyltin; Tri-n-butyl(1-ethoxyvinyl)stannane