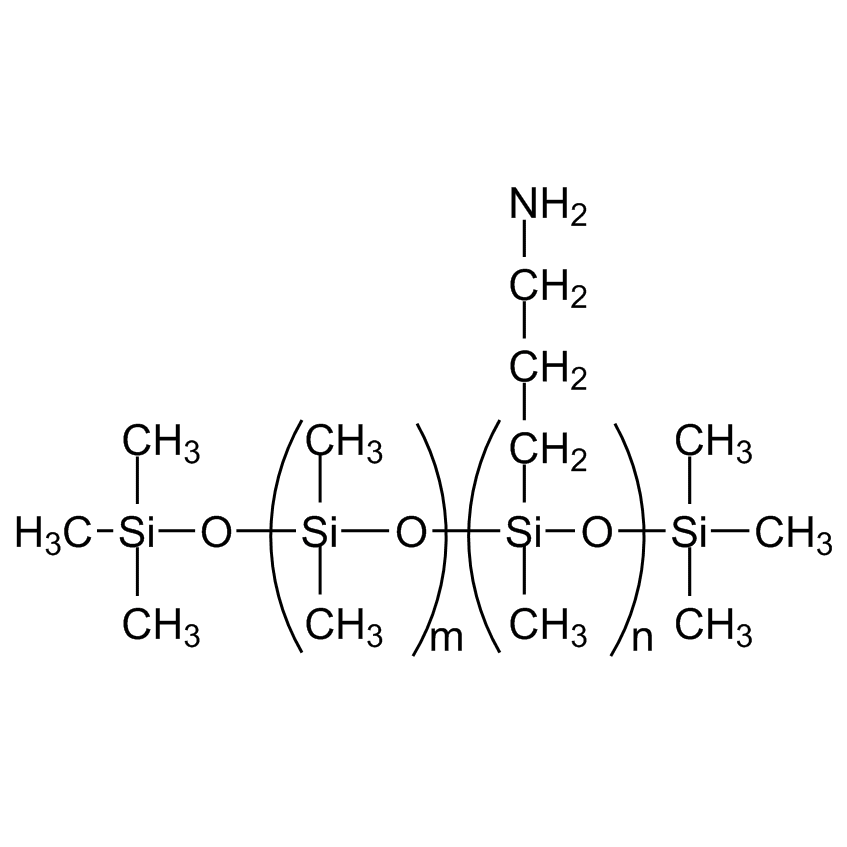Successive ionic layer adsorption and reaction (SILAR) and layer-by-layer (LbL) are thin film fabrication techniques and are increasingly associated with the utilization of silanes and silicones. The films are formed via the deposition of alternating layers of oppositely charged materials with wash steps in between. SILAR typically employs solutions of monomeric ionic materials. Layer-by-layer techniques frequently employ polyions in which two oppositely charged polyions are alternately deposited after intermediate wash steps, W. To prepare SILAR or LbL films with 5 bilayers one would deposit W+W-W+W-W+W-W+W-W+W-W, which would lead to a film with 5 bilayers, specifically +-+-+-+-+-. The bilayers and wash steps can be performed in many different ways including dip coating, spin-coating, spray-coating and flow-based techniques.
SILAR and LbL offer several advantages over other thin film deposition methods. LbL is extremely simple and inexpensive. Another important quality of SILAR and LbL is the high degree of control over layer thickness, which arises due to the linear growth of the films with the number of bilayers. By the fact that each bilayer can be as thin as 1 nm, this method offers facile control over the thickness with 1 nm resolution. Both LbL and SILAR laboratory techniques increasingly use silanes.
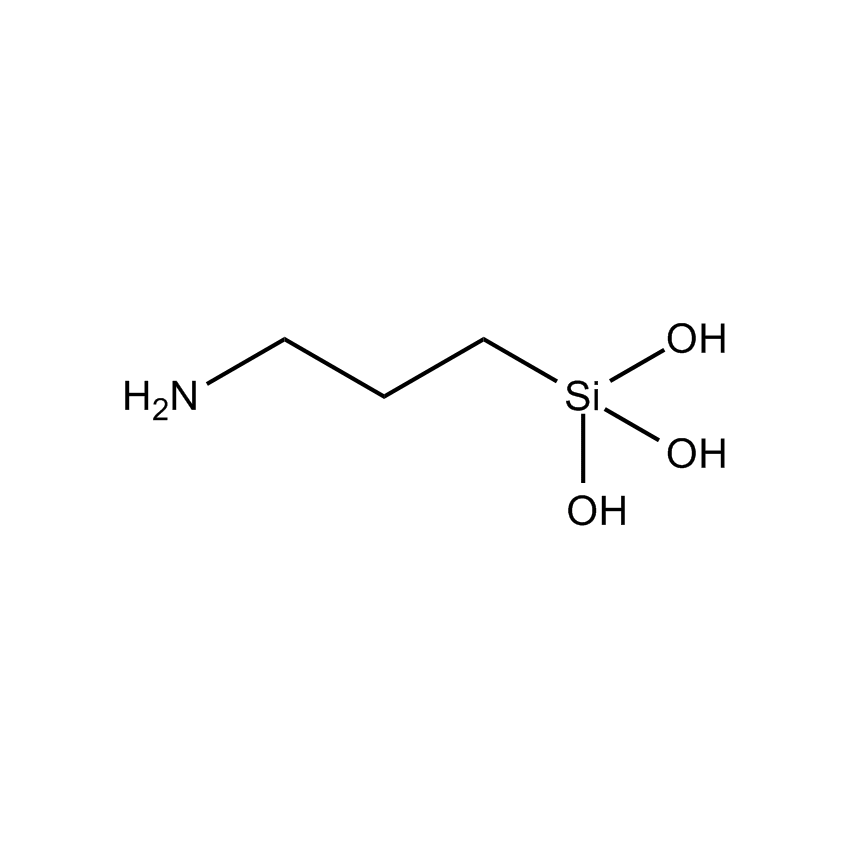
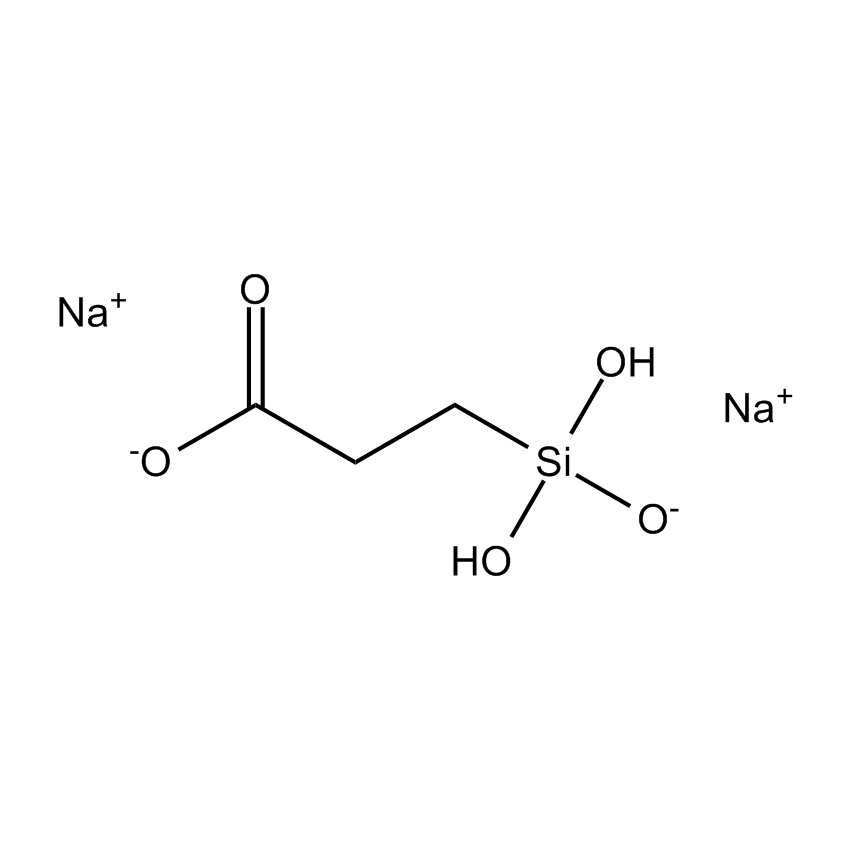
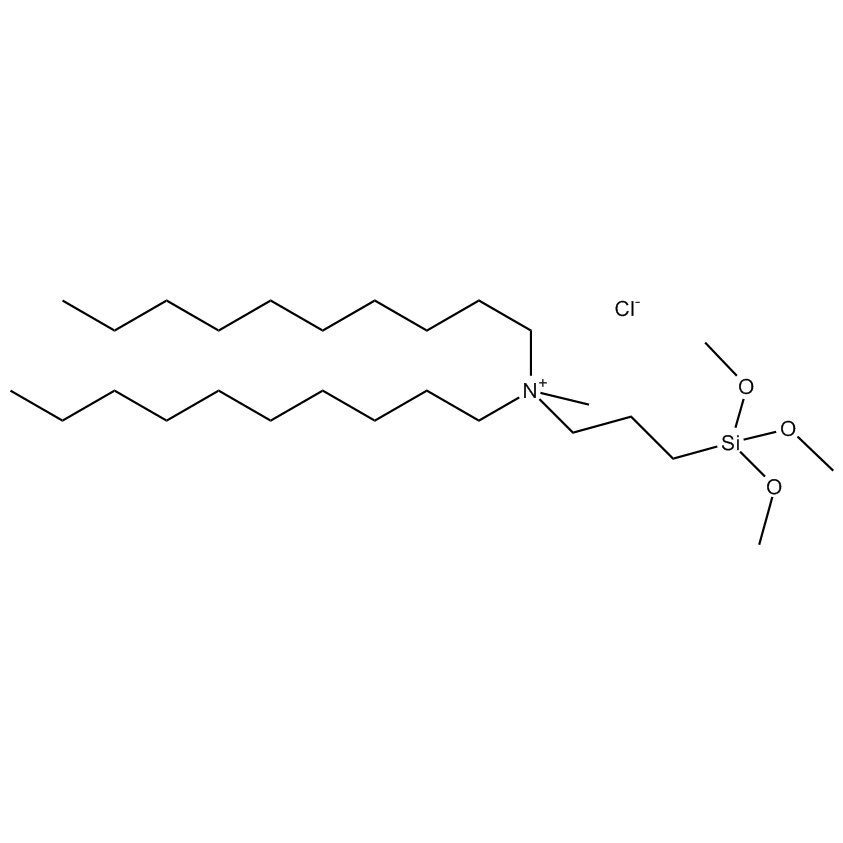
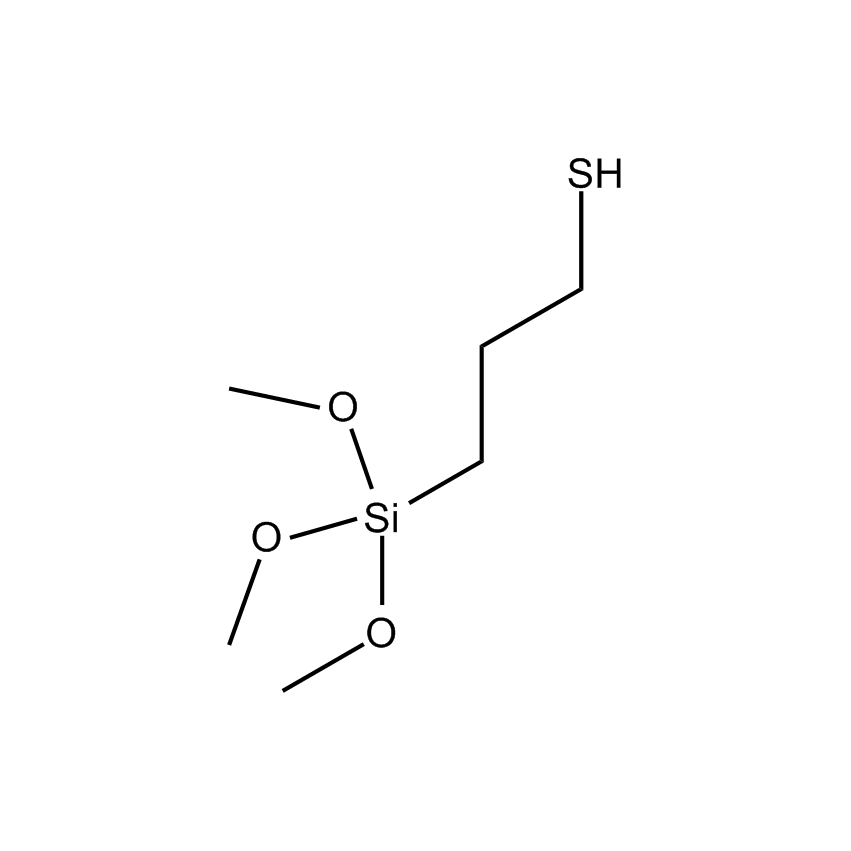
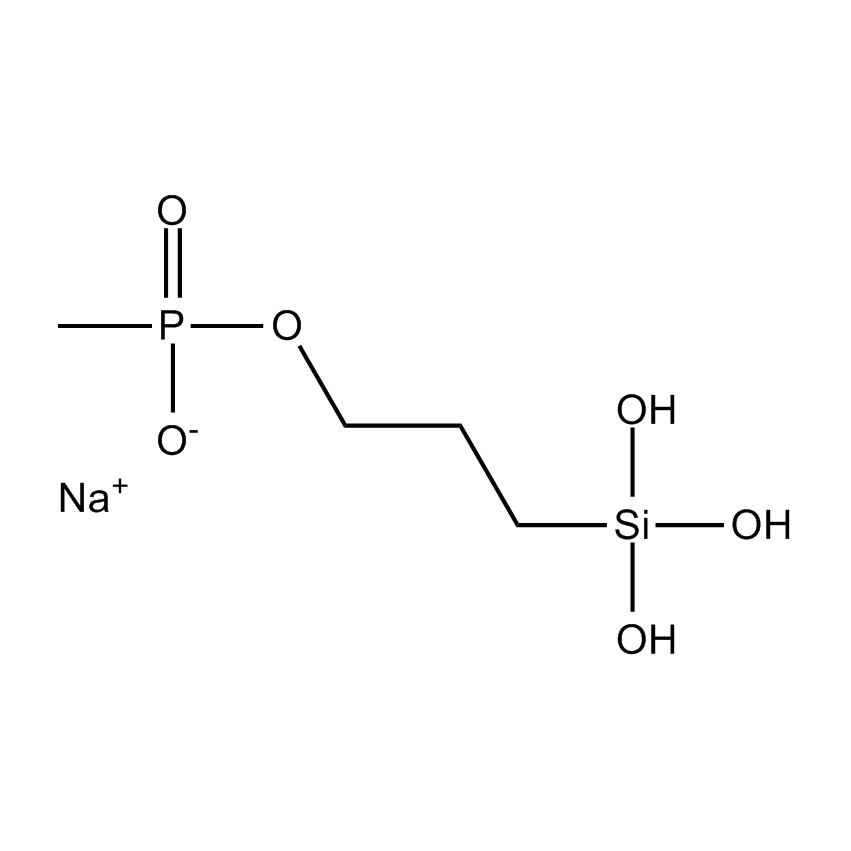
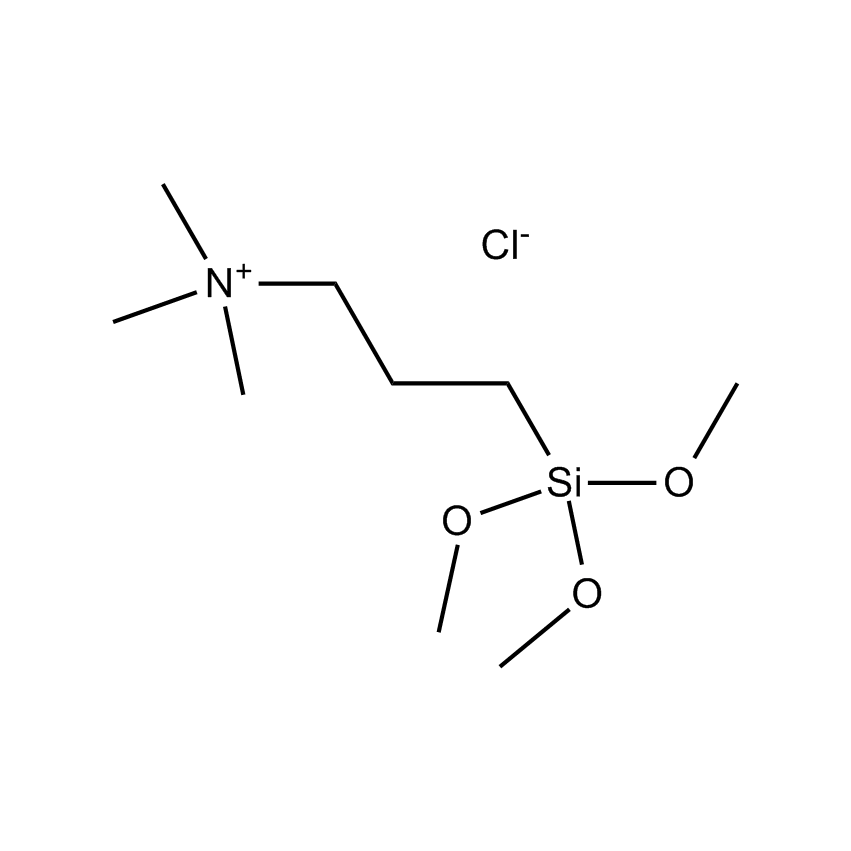
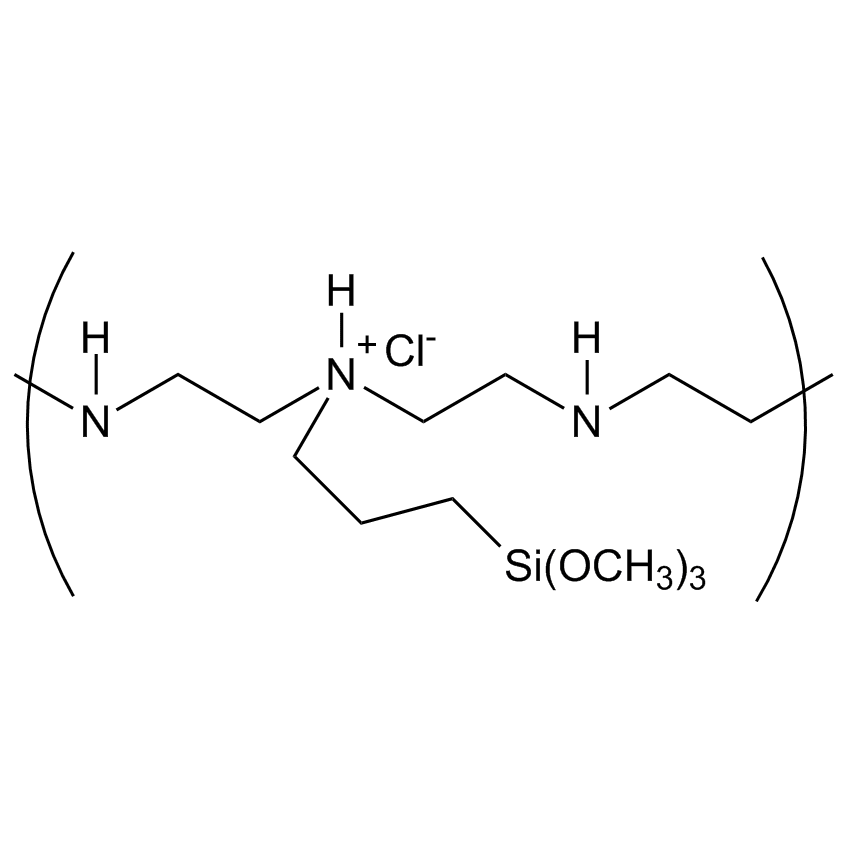
Silanes perform important roles in both SILAR and LbL techniques by modifying the substrate to control deposition. For example, mercaptopropyltrimethoxysilane-modified glass and silica substrates allow SILAR deposition of CdS thin films with alternating cadmium acetate and sodium sulfide. In other instances cationic silanes such as (N-trimethoxysilylpropyl-N,N,N-trimethylammononium chloride) and trimethoxysilylpropyl-modified polyethyleneimine or anionic silanes such as carboxyethylsilanetriol, sodium salt and (3-trihydroxypropylmethylphosphonate, sodium salt form a covalently bound ionic layers on a substrate.
Silicones and silsesquioxanes can also be used directly in SILAR and LbL techniques. Most commonly, amino-functional siloxanes such as AMS-162 or amino-functional silsesquioxanes such as WSA-7011 are utilized directly in the modification of metal substrates or converted to their cationic form by dispersion or dilution in water followed by the stoichiometric addition of hydrochloric acid.