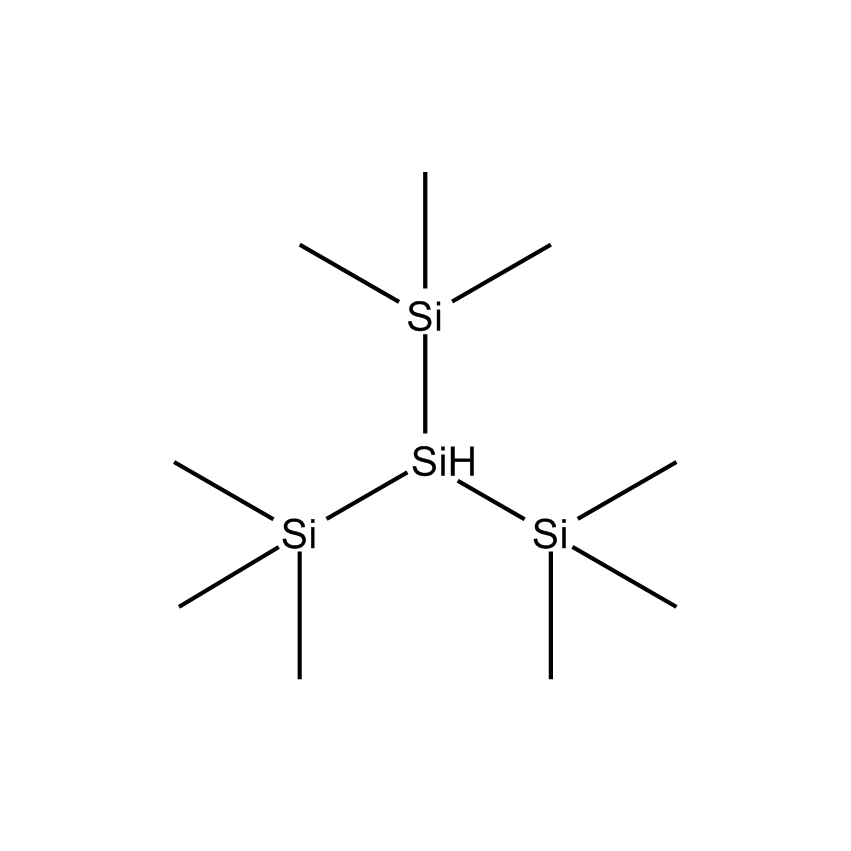
Additional Properties
- Synonyms 1,1,1,3,3,3-HEXAMETHYL-2-TRIMETHYLSILYLTRISILANE; TTMS
- HMIS 2-2-1-X
- Molecular Formula C9H28Si4
- Molecular Weight (g/mol) 248.67
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 82-84/12
- Density (g/mL) 0.806
- Flash Point (˚C) 55 °C
- Refractive Index @ 20˚C 1.489
Application
Reviews.1,2
Efficient mediator in organic radical reactions.3
Initiates addition of alkyl iodides to activated olefins.4
Initiates and promotes the radical addition of perfluoroalkyl iodides to olefins in water.5
Hydrosilylates olefins in aqueous systems in presence of azo initiators.6
Reacts with MeLi to form tris(trimethylsilyl)methyllithium.7
Reacts with LiOMe to form bis(trimethylsilyl)methyllithium.8
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 747-754.
2. Chatgilialoglu, C. Chem. Eur. J. 2008, 14, 2310.
3. Chatgilialoglu, C. Acc. Chem. Res. 1992, 25, 188.
4. Kishimoto, Y.; Ikariya, T. J. Org. Chem. 2000, 65, 7656.
5. Barata-Vallejo, S.; Postigo, A. J. Org. Chem. 2010, 75, 6141.
6. Postigo, A. et al. Organometallics 2009, 28, 3282.
7. Cook, M. A. et al. J. Organomet. Chem. 1970, 24, 529.
8. Sakurai, H. et al. Tetrahedron Lett. 1973, 4193.
Safety
Tri-substituted Silane Reducing Agent
Organosilanes are hydrocarbon-like and possess the ability to serve as both ionic and free-radical reducing agents. These reagents and their reaction by-products are safer and more easily handled and disposed than many other reducing agents. The metallic nature of silicon and its low electronegativity relative to hydrogen lead to polarization of the Si-H bond yielding a hydridic hydrogen and a milder reducing agent compared to aluminum-, boron-, and other metal-based hydrides. A summary of some key silane reductions are presented in Table 1 of the Silicon-Based Reducing Agents brochure.
Tris(trimethylsilyl)silane; 1,1,1,3,3,3-Hexamethyl-2-trimethylsilyltrisilane; TTMS
Silicon Chemistry, Articles
The Si-H bond, based on the relative electrongativities of Si and H, is polarised such that the hydrogen is slightly hydridic in nature. The fact that the silanes are not strongly hydridic makes them excellent candidates for mild and selective reductions of organic functional groups.
Silicon Chemistry, Articles
The reduction of a comprehensive range of organic functional groups ranging from carboxylic acids to aryl fluorides have been shown to be possible with a variety of organosilanes. A comprehensive review of the ionic and transition metal-catalyzed reductions of organic functional groups has recently appeared.
Silicon Chemistry, Articles
Silicon Chemistry, Articles
Some of the general approaches to the key steps in the synthesis of gliflozins, a class of glucose transporters, are discussed. In particular the glycosidation step for the introduction of the key aryl moiety onto the glucose and the reduction steps are presented. Click here for more product information on tetramethyldisiloxane (TMDO, TMDS) reducing agent.