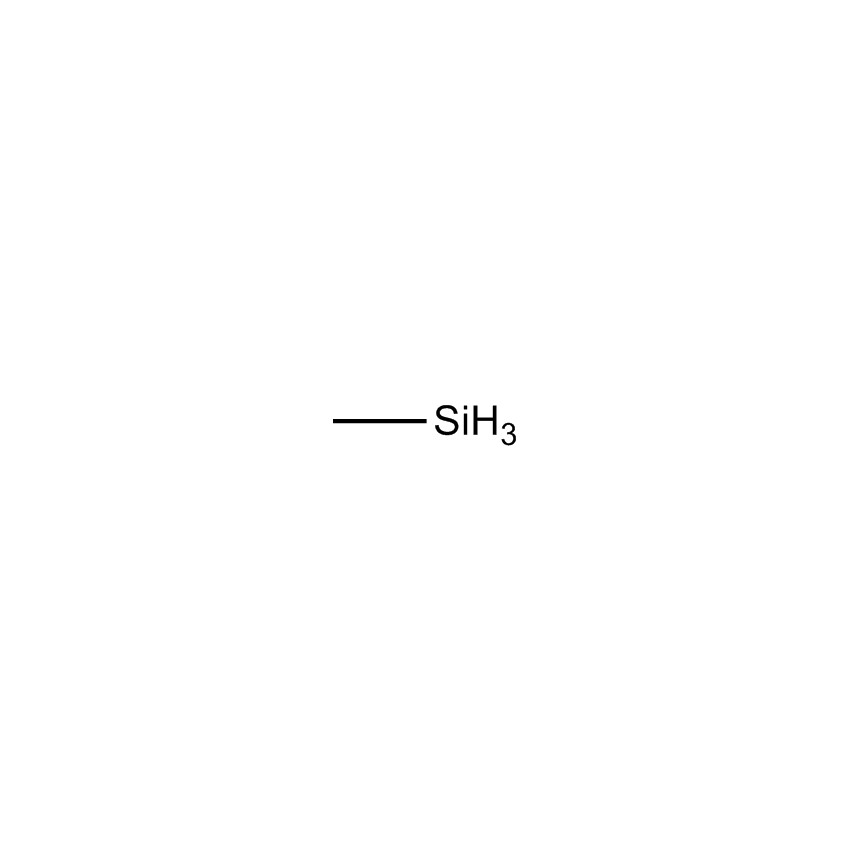Additional Properties
- Einecs Number 213-598-5
- Synonyms 1MS
- HMIS 3-4-3-X
- Molecular Formula CH6Si
- Molecular Weight (g/mol) 46.14
- TSCA Yes
- Delta H Vaporization (kJ/mol) 4.6 kcal/mole
- Autoignition Temp (˚C) 130
- Boiling Point (˚C/mmHg) -57
- Density (g/mL) 0.628
- Density Temperature (˚C) -58
- Melting Point (˚C) -157°
Application
Plasma polymerization yields dry process photoresist.1
Intermediate for poly(methylsilane) precursor to silicon carbide.2
Deposits SiC on Si and Ge at 400 - 500?C.3
Source for hydrogenated amorphous silicon carbide films.4
Reference
1. Dabbagh, G. et al. J. Photopolym. Sci. Tech. 1998, 11, 651.
2. Fhang, Z. et al. J. Am. Ceram. Soc. 1991, 74, 670.
3. Takatsuka, T. et al. Appl. Surf. Sci. 2000, 162, 156.
4. Lee, M. et al. in “Chemical Aspects of Electronic Ceramics Processing” Arkles, B. ed., MRS Proc. 1998, 495, 153.
Safety
Volatile Carbosilane
Carbosilanes are compounds in which the elements of silicon and carbon alternate in a molecular framework or polymeric backbone in an approximate ratio of 1:1. By appropriate selection of the carbosilane precursor and deposition conditions the silicon carbide framework can be shifted toward substituted silicon and diamond-like structures.
Methylsilane; 1MS; Silylmethane; Monomethylsilane; Monosilylmethane
CAUTION: CAN FORM EXPLOSIVE MIXTURES WITH AIR
Silicon Chemistry, Applied Technology, Metal-Organic Chemistry, Applied Chemistry & Physics
Silicon Chemistry, Applied Technology
Silicon Chemistry, Applied Technology, Metal-Organic Chemistry, Applied Chemistry & Physics
Silicon Chemistry, Applied Technology