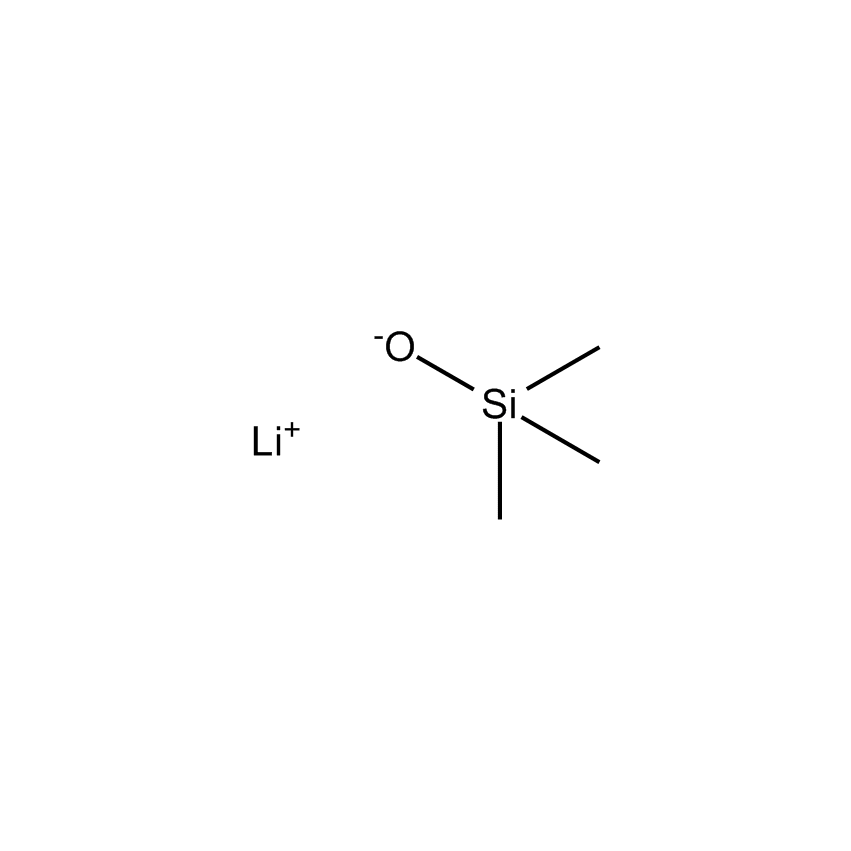Energy storage plays a crucial part of our everyday lives and lithium-ion batteries have become the battery type of choice in most applications due to the high energy density of this technology compared to other battery technologies such as nickel-type batteries and lead acid batteries. The market for lithium-ion batteries is projected to grow significantly in both consumer electronics and automotive. Key drivers in the growth of lithium-ion batteries include the increased growth of hybrid electric and all-electric vehicles, as well as the overall growth in portable electronic devices. Lithium metal batteries have potential to be even better than lithium-ion batteries.
Lithium-ion batteries are composed of a cathode, anode, electrolyte, and separator. Cathode materials include lithium iron phosphate (LiFePO4), lithium manganese oxide (LiMn2O4), Lithium cobalt oxide (LiCoO2), lithium nickel cobalt manganese oxide (also known as “NMC”) (LiNiMnCoO2), lithium nickel cobalt aluminum oxide (LiNiCoAlO2), and lithium titanate. Anode materials are typically graphite. Separators are typically microporous polypropylene or polyethylene film. And electrolytes are typically lithium hexafluorophosphate (LiPF6) in ethylene carbonate (EC).
Materials development in lithium-ion batteries is being driven by the need for improved battery capacity, lower cost (for automotive applications), improved safety, and improved capacity retention after cycling of the battery. Improvements in battery capacity and cost are being addressed through modification of electrode materials and electrode thickness, while improvements in safety and capacity retention are being addressed by modification of the electrolytes through additives and entirely new electrolyte materials.
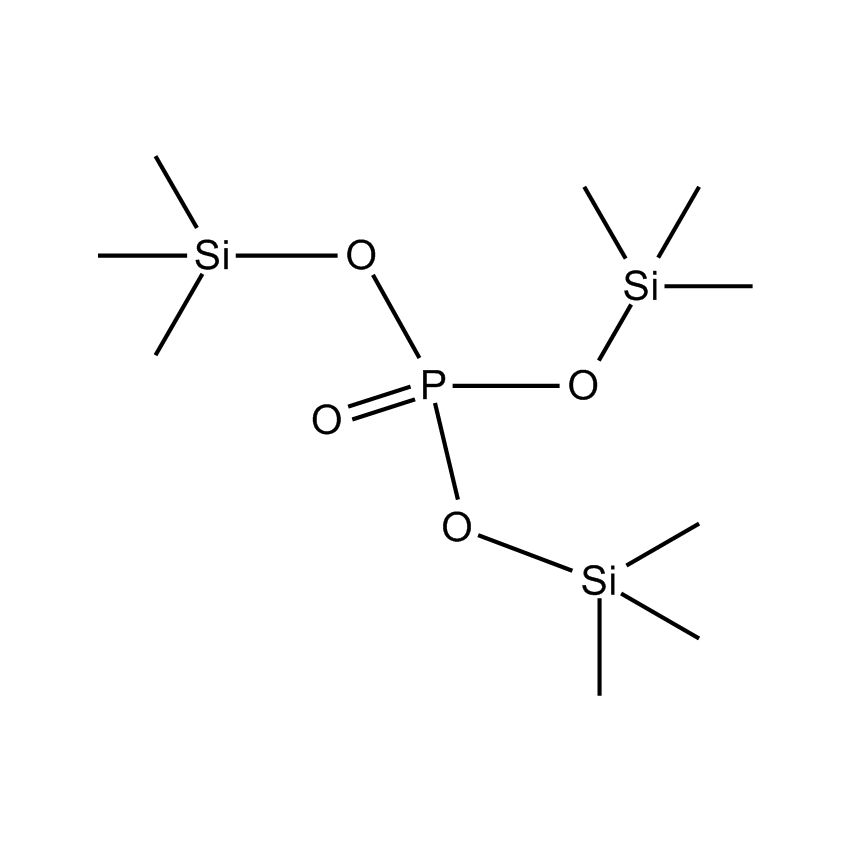
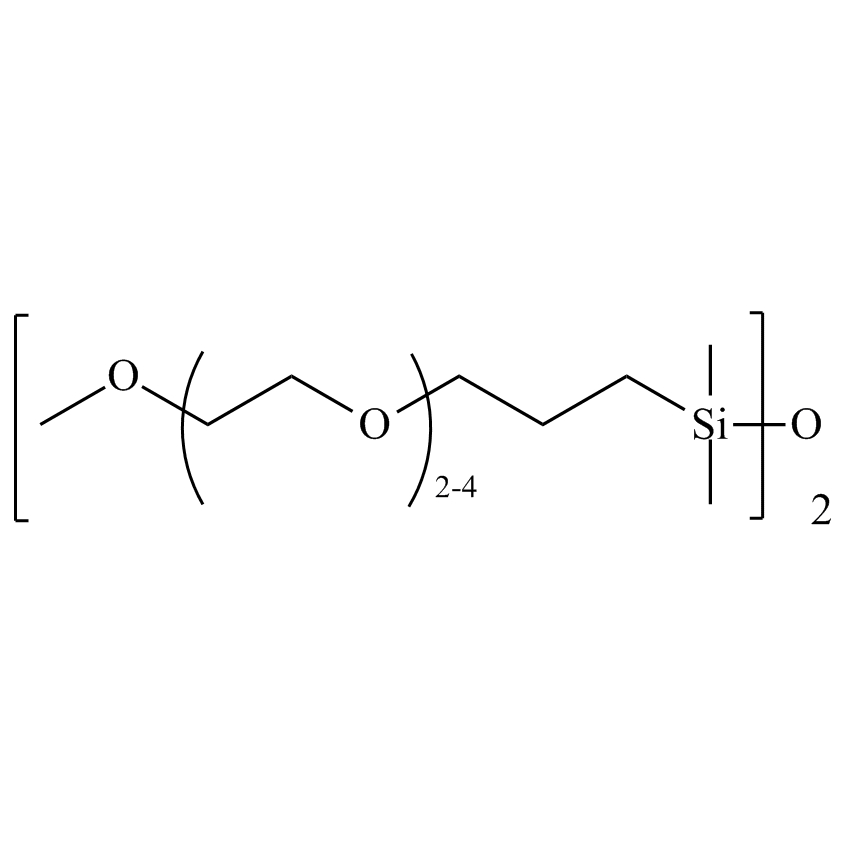
Silanes can improve electrolyte stability. Silyl phosphates such as tris(trimethylsilyl)phosphate (SIT8723.0) have been used as additives in the ethylene carbonate electrolyte to improve stability and capacity retention (US 8,308,971). Polyether-functionalized silanes have also been developed as improved electrolytes (Chen, X., et al, Journal of Power Sources 241 (2013) 311-319). Recently, dimethylaminoethoxyalkyl silanes have also been developed as electrolyte additives to improve stability and capacity retention (Wang, J.L., et al. Science China Chemistry, 2013, 56(6), 739-745). Furthermore, the use of bis(methoxytriethyleneoxypropyl)-tetramethyldisiloxane (SIB1550.0) allows for a high lithium content to be established as the small siloxane unit inhibits the polyethylene glycol chain from crystallizing, enabling a high lithium content to be established at an electrode interface.
Uses
Mobile phones and laptop computers
both depend on long lasting batteries to allow us to work and live efficiently.
Automobiles
cars are relying on improved batteries as a secondary power source to gasoline and as the sole energy source in full electric vehicles.
Solar energy
solar energy is made more economical by storage of energy in batteries.