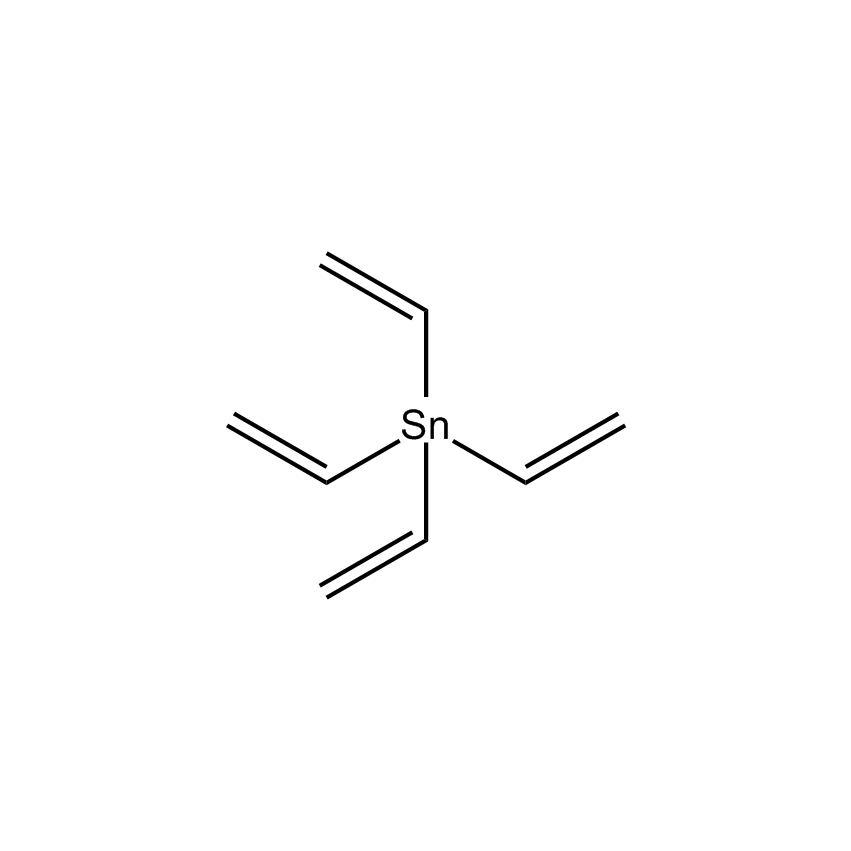
Additional Properties
- Einecs Number 214-193-6
- HMIS 3-3-0-X
- Molecular Formula C8H12Sn
- Molecular Weight (g/mol) 226.88
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 160-163
- Density (g/mL) 1.257
- Flash Point (˚C) 40 °C
- Refractive Index @ 20˚C 1.4914
Application
Reviewed.1
Asymmetrically allylates prochiral ketones with high ee's.2
Allylates ?-keto esters with high enantioselectivty.3
Reference
1. Scott, W. J.; Moretto, A. F. in Encyclopedia of Reagents for Organic Synthesis 1995, Volume 7, 4830-4831.
2. Wooten, A. J. et al. Org. Lett. 2007, 9, 381.
3. Zheng, K. et al. J. Org. Chem. 2007, 72, 8478.
Safety
Organotin Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Tetravinyltin; Tetraethenylstannane
Metal-Organic Chemistry, Articles
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products