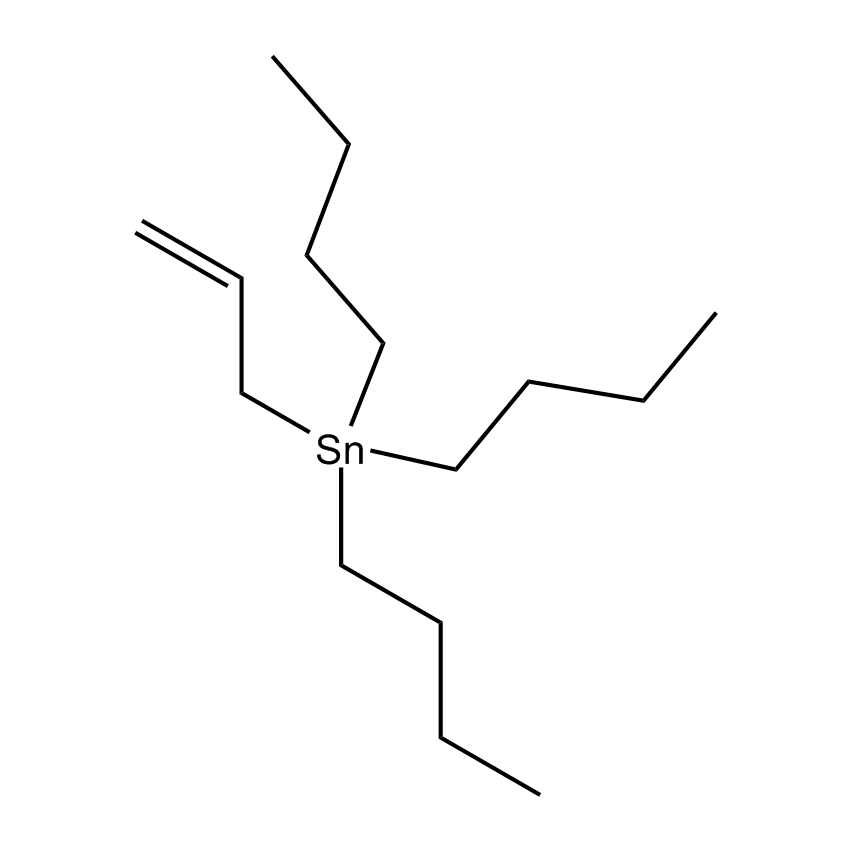
Additional Properties
- Einecs Number 246-494-3
- HMIS 2-2-1-X
- Molecular Formula C15H32Sn
- Molecular Weight (g/mol) 331.11
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 88-92/0.2
- Density (g/mL) 1.068
- Flash Point (˚C) 103 °C
- Refractive Index @ 20˚C 1.4846
Application
Reviewed.1
Conjugatively allylates alkylidene Meldrum’s acids.10
Generates homoallylic amines with aldehydes and amines.11
Allylates bromides and chloroethers.,2,3
Radical initiation induces 1,2-addition of allyltins and alkyl iodides to electron deficient alkenes.4
Polymerization by Natta catalyst - Et3Al-TiCl4.5
Converts aldehydes to homoallylic alcohols and imines to homoallylic amines.6
Converts combination of aldehyde and amine to homoallylic amines.7
Enantioselectively generates homoallylic amines in a 3-component reaction scheme.8
Adds enantioselectively to prochiral ketones to form homoallyl alcohols in good enantioselectively.9
Fieser
F&F: Vol. 9, p 8; Vol. 11, p 15; Vol. 12, p 21; Vol. 13, p. 10; Vol. 14, p. 14; Vol. 16, p. 7; Vol. 17, p 12.
Reference
1. Castellino, S. in Encyclopedia of Reagents for Organic Synthesis 1995, Volume 1, 125-128.
10. Duma, A. N.; Fillion, E. Org. Lett. 2009, 11, 1919.
11. Narsaiah, A. et al. Synthesis, 2010, 1609.
2. Corey, E. et al. Tetrahedron Lett. 1990, 31, 3715.
3. Keck, G. et al. J. Am. Chem. Soc. 1982, 104, 5829.
4. Mizuno, K. et al. F&F: Vol. 14, p 16; J. Am. Chem. Soc. 1988, 110, 1288.
5. Montecatini. Soc. Chem. Abstr. 53, 1837; Ital. Patent 589,299, 1959.
6. Shen, W. et al. Tetrahedron Lett. 2008, 49, 4047.
7. Kalita, P. K.; Phukan, P. Tetrahedron Lett. 2008, 49, 5495.
8. Li, X. et al. Chem. Eur. J. 2008 14, 4796.
9. Lu, J. et al. Tetrahedron Lett. 2005, 46, 7435.
Safety