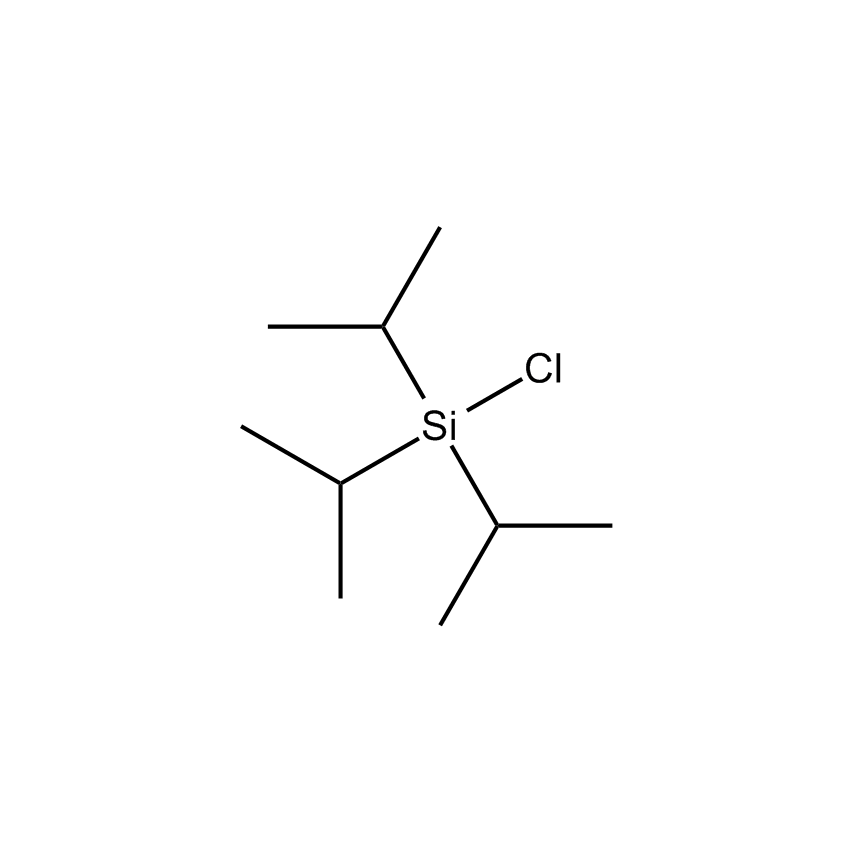
Additional Properties
- Einecs Number 603-492-0
- Synonyms TIPS-CHLORIDE
- HMIS 3-2-1-X
- Molecular Formula C9H21ClSi
- Molecular Weight (g/mol) 192.81
- Purity (%) 97%
- TSCA No
- Autoignition Temp (˚C) 255
- Boiling Point (˚C/mmHg) 79-80/10
- Density (g/mL) 0.903
- Flash Point (˚C) 62 °C
- Refractive Index @ 20˚C 1.4520
Application
Review of synthetic utility.1
Review on organosilane protecting groups.2
Employed in nucleotide synthesis.3
Protection and stereocontrol of reaction of pyrroles.4
Quantitatively converts O-acetyl-protected glycosylthiomethyl phosphonate esters to their phosphonates.5
Used to form stable enol silyl ethers that can be oxidized to enones or ?-TIPS-enones.6
Reacts with hydrazine to form the mono-TIPShydrazine, which can be converted to TIPS-protected hydrazones and further to unsymmetrical azines.7
Fieser
F&F: Vol. 5, p 75.
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 554-558.
2. Larson, G. L. “Silicon-Based Blocking Agents” Gelest, Inc. 2014.
3. Ogilvie, K. et al. Tetrahedron Lett. 1974, 2861. Ogilvie, K. et al. Carbohyd., Nucleosides, Nucleotides 1976, 3, 197.
4. Pattenden, G.; Robertson, G. M. Tetrahedron Lett. 1986, 27, 399.
5. Zhu, X. et al. J. Org. Chem. 2004, 69, 6843.
6. Yu, J.-Q. et al. Org. Lett. 2005, 7, 1415.
7. Justo de Pomar, J. C.; Soderquist, J. A. Tetrahedron Lett. 2000, 41, 3285.
Safety
Trialkylsilyl Blocking Agent
Used as a protecting group for reactive hydrogens in alcohols, amines, thiols, and carboxylic acids. Organosilanes are hydrogen-like, can be introduced in high yield, and can be removed under selective conditions. They are stable over a wide range of reaction conditions and can be removed in the presence of other functional groups, including other protecting groups. The tolerance of silylated alcohols to chemical transformations summary is presented in Table 1 of the Silicon-Based Blocking Agents brochure.
Triisopropylchlorosilane; Chlorotriisopropylsilane; TIPS-Cl