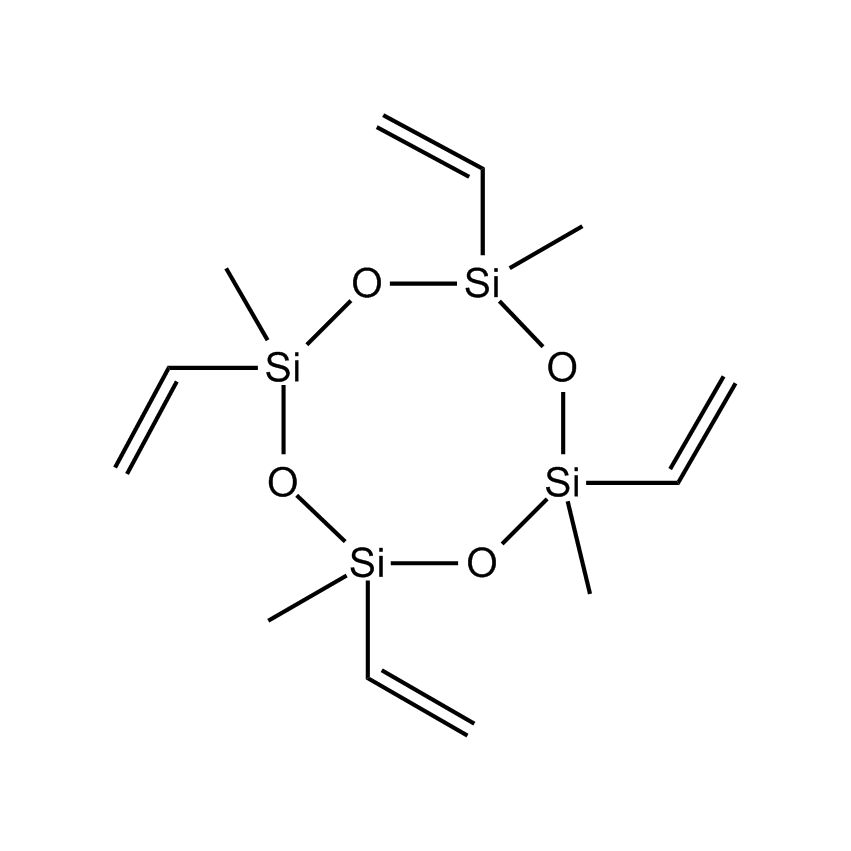
Additional Properties
- Einecs Number 219-863-1
- HMIS 2-1-0-X
- Molecular Formula C12H24O4Si4
- Molecular Weight (g/mol) 344.66
- Purity (%) 97%
- TSCA Yes
- Boiling Point (˚C/mmHg) 110/10
- Density (g/mL) 0.998
- Flash Point (˚C) 102 °C
- Melting Point (˚C) -43°
- Refractive Index @ 20˚C 1.4342
- Viscosity at 25 ˚C (cSt) 3.9
Application
Review of synthetic utility.1
Modifier for Pt-catalyst in 2-component RTVs.2
Core molecule for dendrimers.3
Excellent and inexpensive reagent for vinylations in cross-coupling reactions for the formation of styrenes and dienes.4,5,6
Extensive review on the use in silicon-based cross-coupling reactions.7
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 484-487.
2. Kim, C. et al. Bull. Kor. Chem. Soc. 1997, 18, 164.
3. Broadbridge, R. et al. J. Chem. Soc., Chem. Commun. 1998, 1449.
4. Denmark, S. E.; Butler, C. R. Org. Lett. 2006, 8, 63.
5. Denmark, S. E.; Wang, Z. J. Organomet. Chem. 2001, 624, 372.
6. Denmark, S. E.; Wehrli, D. Org. Lett. 2000, 2, 565.
7. Denmark, S. E. et al. Organic Reactions, Vol. 75, Denmark, S. E. ed., John Wiley and Sons, 233, 2011.
Safety
Alkenylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
1,3,5,7-Tetravinyl-1,3,5,7-tetramethylcyclotetrasiloxane; Methylvinylcyclosiloxane; Tetramethyltetravinylcyclotetrasiloxane; Tetramethyltetraethenylcyclotetrasiloxane
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products