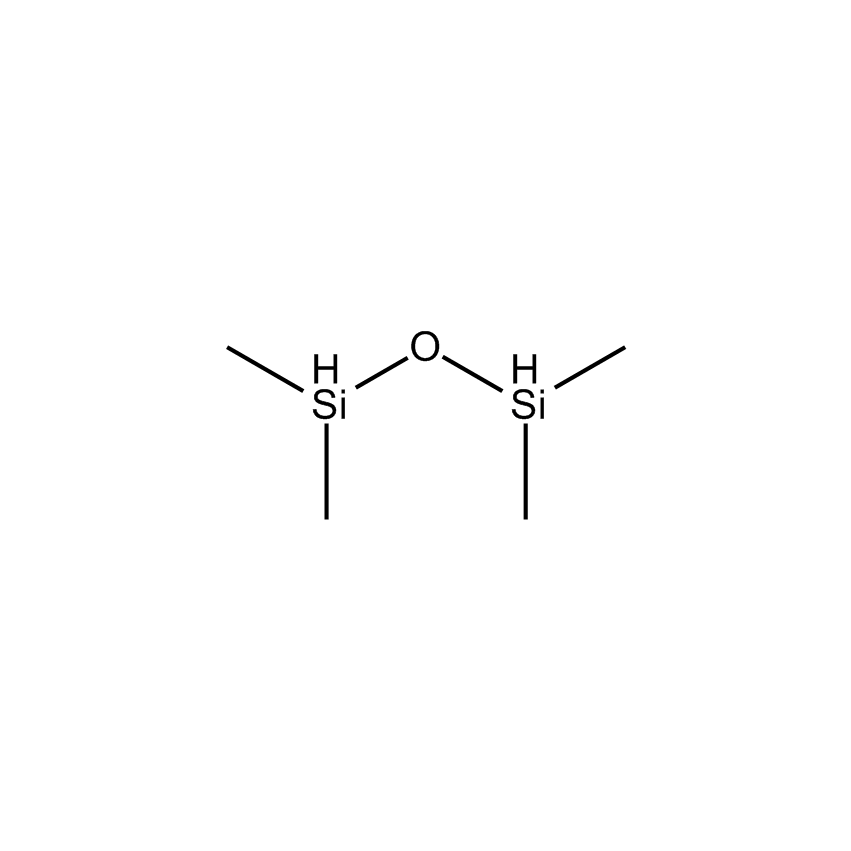
Additional Properties
- Einecs Number 221-906-4
- HMIS 2-4-1-X
- Molecular Formula C4H14OSi2
- Molecular Weight (g/mol) 134.22
- Purity (%) 98%
- TSCA Yes
- Delta H Vaporization (kJ/mol) 7.25 kcal/mole
- Autoignition Temp (˚C) 208
- Boiling Point (˚C/mmHg) 70-71
- Density (g/mL) 0.757
- Flash Point (˚C) -12 °C
- Refractive Index @ 20˚C 1.3669
- Additional Viscosity (cSt) '0.56, 20
- Viscosity at 25 ˚C (cSt) 0.56
Application
Employed in reductive halogenation of aldehydes and epoxides.1
Used to link ferrocenylsilane, polyolefin block copolymers into stable cylindrical forms.2
Employed in the high-yield reduction of amides to amines in the presence of other reducible groups.3
Reduces anisoles to arenes.4
Hydrosilylates terminal alkynes to form alkenylsilanes capable of cross-coupling w/ aryl and vinyl halides.5
Employed in the reduction of an acetopheone derivative to the methylene in the synthesis of ziprasidone.6
Reference
1. Azipura et al. Tetrahedron Lett. 1984, 25, 3123.
2. Wang, X.-S. et al. J. Am. Chem. Soc. 2003, 125, 12686.
3. Hanada, S. et al. J. Am. Chem. Soc. 2009, 131, 15032.
4. Alvarez-Bercedo, P.; Martin, R. J. Am. Chem. Soc. 2010, 132, 17352.
5. Denmark, S. E.; Wang, Z. Org. Lett. 2001, 3, 1073.
6. Nadkami, D.; Hallissey, J. F. Org. Proc. Res. Dev. 2008, 12, 1142.
Safety
Alkenylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
ALD Material
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
Siloxane-Based Silane Reducing Agent
Organosilanes are hydrocarbon-like and possess the ability to serve as both ionic and free-radical reducing agents. These reagents and their reaction by-products are safer and more easily handled and disposed than many other reducing agents. The metallic nature of silicon and its low electronegativity relative to hydrogen lead to polarization of the Si-H bond yielding a hydridic hydrogen and a milder reducing agent compared to aluminum-, boron-, and other metal-based hydrides. A summary of some key silane reductions are presented in Table 1 of the Silicon-Based Reducing Agents brochure.
1,1,3,3-Tetramethyldisiloxane; 1,1-Dihydro-1,1,3,3-tetramethyldisiloxane; TMDO; TMDS
Silicon Chemistry, Articles
The Si-H bond, based on the relative electrongativities of Si and H, is polarised such that the hydrogen is slightly hydridic in nature. The fact that the silanes are not strongly hydridic makes them excellent candidates for mild and selective reductions of organic functional groups.
Silicon Chemistry, Articles
The reduction of a comprehensive range of organic functional groups ranging from carboxylic acids to aryl fluorides have been shown to be possible with a variety of organosilanes. A comprehensive review of the ionic and transition metal-catalyzed reductions of organic functional groups has recently appeared.
Silicon Chemistry, Articles
Silicon Chemistry, Articles
Some of the general approaches to the key steps in the synthesis of gliflozins, a class of glucose transporters, are discussed. In particular the glycosidation step for the introduction of the key aryl moiety onto the glucose and the reduction steps are presented. Click here for more product information on tetramethyldisiloxane (TMDO, TMDS) reducing agent.