Additional Properties
- HMIS 3-2-1-X
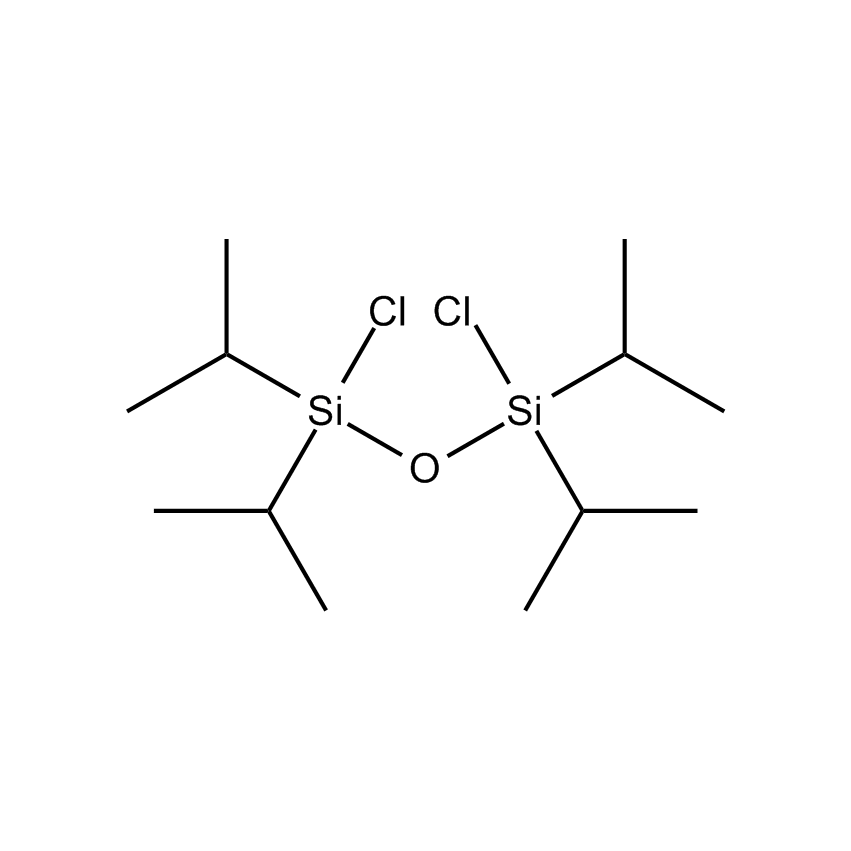
- Molecular Formula C12H28Cl2OSi2
- Molecular Weight (g/mol) 315.43
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 120/1.5
- Density (g/mL) 0.986
- Flash Point (˚C) 76 °C
- Refractive Index @ 20˚C 1.4543
Application
Review of synthetic utility.1
Review on organosilane protecting groups.2
Reagent for protection of 3',5' hydroxy nucleosides.3,4
Key review in natural products synthesis.5
Protects the four equatorial hydroxyls of inositol in a single step.6
Fieser
F&F: Vol. 16, p 125.
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 230-233.
2. Larson, G. L. “Silicon-Based Blocking Agents” Gelest, Inc. 2014.
3. Kanaya, E. N. et al. Biochemistry 1987, 26, 7159.
4. Markiewicz, W. J. Chem. Res. 1979, 24.
5. Ziegler, T. et al. Trends Org. Chem. 1997, 6, 91.
6. Martin-Lomas, M. et al. Eur. J. Org. Chem. 2000, 1539.
Safety
Bridging Silicon-Based Blocking Agent
Used as a protecting group for reactive hydrogens in alcohols, amines, thiols, and carboxylic acids. Organosilanes are hydrogen-like, can be introduced in high yield, and can be removed under selective conditions. They are stable over a wide range of reaction conditions and can be removed in the presence of other functional groups, including other protecting groups. The tolerance of silylated alcohols to chemical transformations summary is presented in Table 1 of the Silicon-Based Blocking Agents brochure.
Tetraisopropyldichlorodisiloxane; Dichlorotetraisopropyldisiloxane; 1,3-Dichloro-1,1,3,3-tetraisopropyldisiloxane; TIPDS