Additional Properties
- Einecs Number 233-054-0
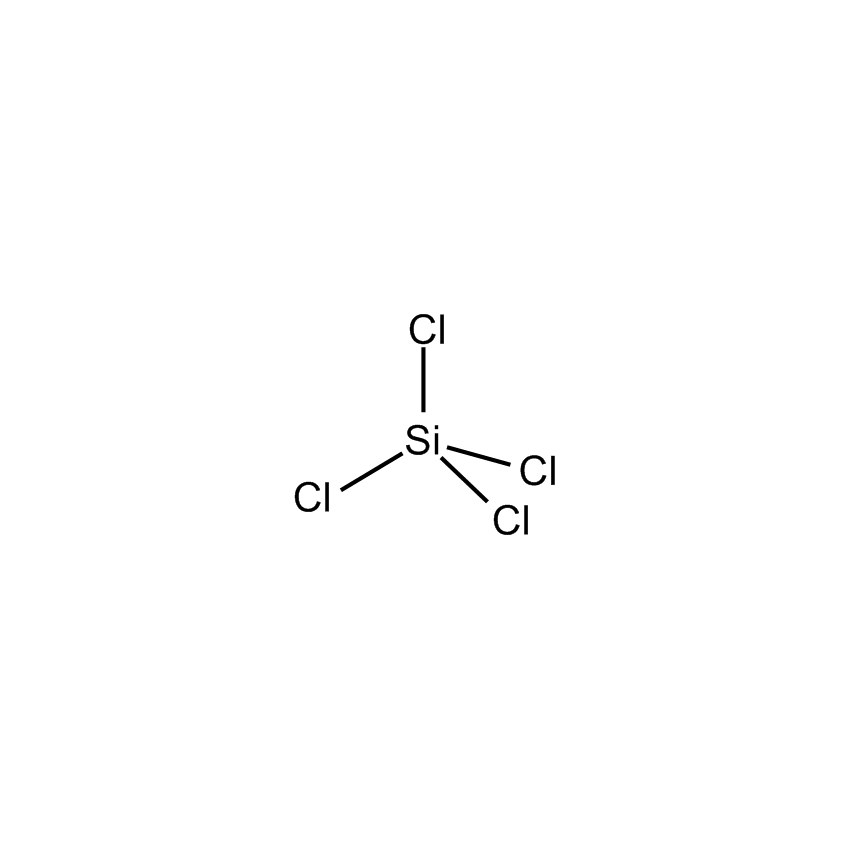
- Synonyms SILICON TETRACHLORIDE
- HMIS 3-0-2-X
- Molecular Formula Cl4Sn
- Molecular Weight (g/mol) 169.90
- Purity (%) 98%
- TSCA Yes
- Delta H Vaporization (kJ/mol) 7.6 kcal/mole
- Boiling Point (˚C/mmHg) 57.6
- Density (g/mL) 1.481
- Melting Point (˚C) -70°
- Refractive Index @ 20˚C 1.4153
- Viscosity at 25 ˚C (cSt) 0.35
Application
Enantioselectively opens stilbine epoxides to trichlorosilylated chlorohydrins.1
Promotes the reaction of aldehydes with isocyanides.2
Reference
1. Tao, B. et al. J. Am. Chem. Soc. 2001, 123, 353.
2. Denmark, S. E.; Fan, Y. J. Am. Chem. Soc. 2003, 125, 7825.
Safety
ALD Material
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
Tetrachlorosilane; Silicon chloride; Silicon tetrachloride