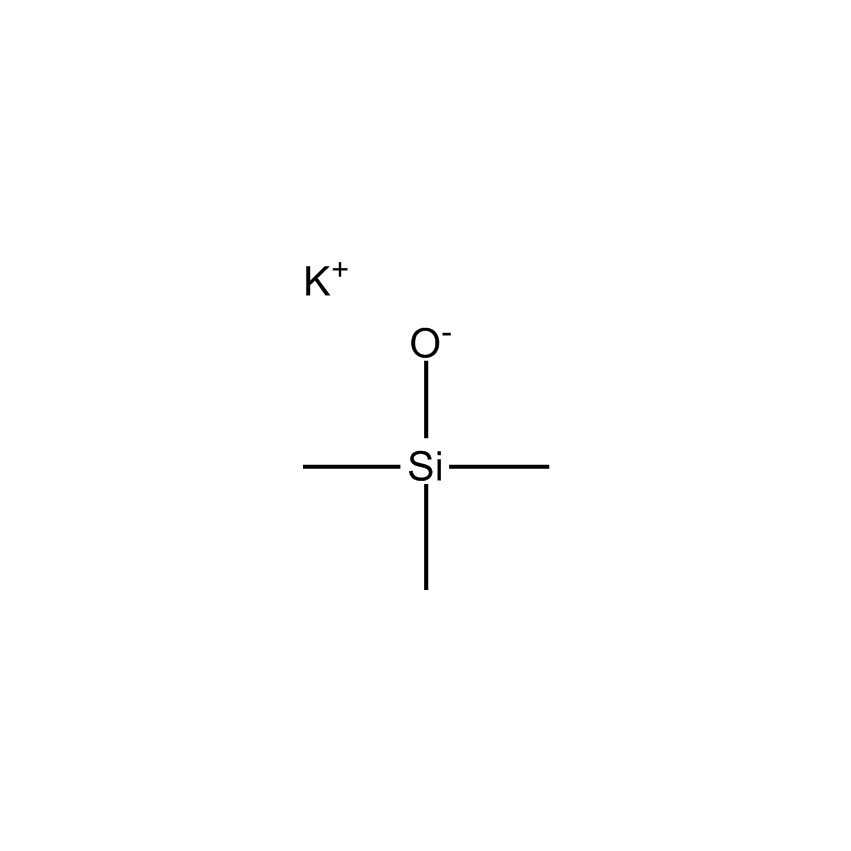
Additional Properties
- Einecs Number 234-062-7
- HMIS 3-2-1-X
- Molecular Formula C3H9KOSi
- Molecular Weight (g/mol) 128.29
- Purity (%) 95%
- TSCA Yes
- Boiling Point (˚C/mmHg) NA
- Melting Point (˚C) 134-8° dec
Application
Cleaves esters under non-aqueous conditions.1
Catalyzes fluoride-free, silicon-based cross-coupling reactions.2
Converts difluorobenzenes to fluorophenols.3
Catalyst for the cross-coupling of aryl and vinylsilanes.4
Fieser
F&F: Vol. 12, p 414.
Reference
1. Laganis, E. et al. Tetrahedron Lett. 1984, 25, 5831.
2. Denmark, S. E.; Regens, C. S. Accts. Chem. Res. 2008, 41, 1486.
3. Li, J. et al. Synlett 2009, 633.
4. Jeffery, T. Tetrahedron Lett. 1999, 40, 1673.
Safety
Silane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Potassium trimethylsilanolate; Potassium trimethylsiloxide; Trimethylsilanol, potassium salt
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products