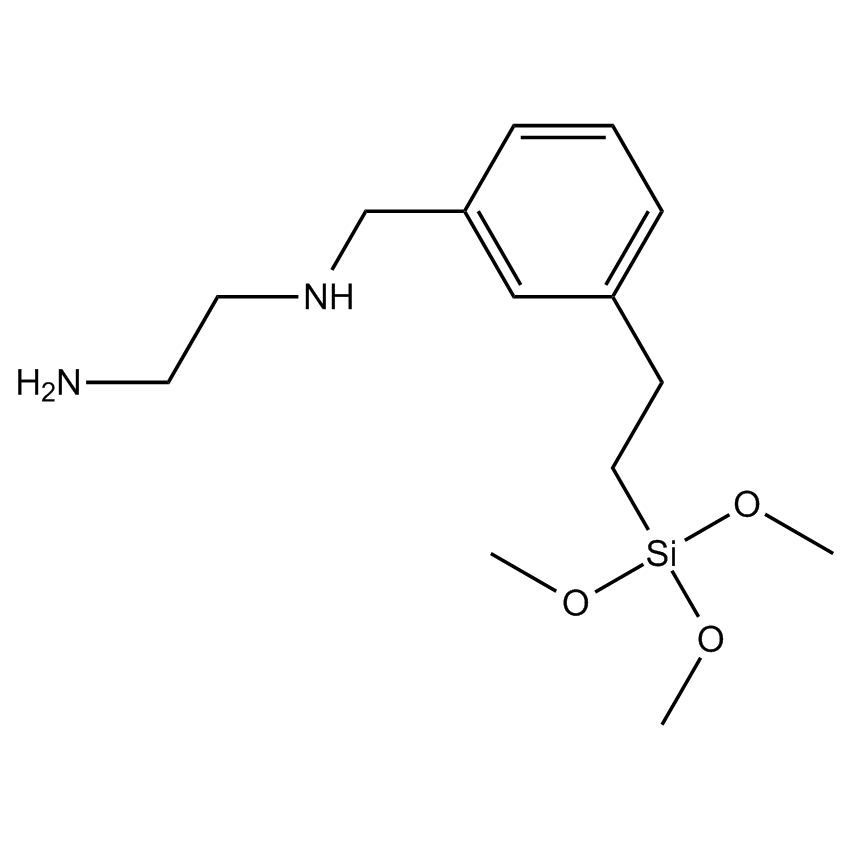
Additional Properties
- HMIS 3-1-1-X
- Molecular Formula C14H26N2O3Si
- Molecular Weight (g/mol) 298.46
- Purity (%) 80% mixed m,p isomers
- TSCA Yes
- Boiling Point (˚C/mmHg) 126-130/0.2
- Density (g/mL) 1.02
- Flash Point (˚C) >110 °C
- Refractive Index @ 20˚C 1.5083
Application
Coupling agent for polyimides.1
Photochemically sensitive (194nm).2
Forms self assembled monolayers.3
Reagent for charge heterogeneity in micropatterning.4
Reference
1. Arkles, B. et al. Modern Plastics 1980, 57, 64.
2. Dressick, W. et al. Thin Solid Films 1996, 284, 568.
3. Harnett, C. et al. Appl. Phys. Lett. 2000, 76, 2466.
4. Chen, J. et al. Nano Lett. 2002, 2, 393.
Safety
Diamino Functional Trialkoxy Silane
Silane coupling agents have the ability to form a durable bond between organic and inorganic materials to generate desired heterogeneous environments or to incorporate the bulk properties of different phases into a uniform composite structure. The general formula has two classes of functionality. The hydrolyzable group forms stable condensation products with siliceous surfaces and other oxides such as those of aluminum, zirconium, tin, titanium, and nickel. The organofunctional group alters the wetting or adhesion characteristics of the substrate, utilizes the substrate to catalyze chemical transformations at the heterogeneous interface, orders the interfacial region, or modifies its partition characteristics, and significantly effects the covalent bond between organic and inorganic materials.
(Aminoethylaminomethyl)phenethyltrimethoxysilane, [N-(2-Aminoethyl)aminomethylphenyl]ethyltrimethoxysilane, N-1-[[[2-(Trimethoxysilyl)ethyl]phenyl]methyl-1-2-ethanediamine
Silicon Chemistry, Articles
APPLYING A SILANE COUPLING AGENT NOTE — Product codes in the tables are not links. Please use the Product Search functions to obtain detailed information about these products. Deposition from aqueous alcohol solutions is the most facile method for preparing silylated surfaces. A 95% ethanol / 5% water solution is adjusted to pH 4.5–5.5 with acetic acid. Silane is added with stirring to yield a 2% final concentration. Five minutes should be allowed for hydrolysis and silanol formation. Large objects, e.g. glass plates, are dipped into the solution, agitated gently, and removed after 1–2 minutes. They are rinsed free of excess materials by dipping briefly in ethanol. Particles, e.g. fillers and supports, are silylated by stirring them in solution for 2–3 minutes and then decanting the solution. The particles are usually rinsed twice briefly with ethanol. Cure of the silane layer is for 5–10 minutes at 110°C or 24 hours at room temperature (60% relative humidity). For aminofunctional silanes such as SIA0610.0 and SIA0589.0 this procedure is modified by omitting the addition of acetic acid. The procedure is not acceptable for chlorosilanes as bulk polymerization often occurs. Silane concentration of 2% is a starting point. It usually results in deposition of trialkoxysilanes as 3–8 molecular layers. Monoalkoxysilanes are always deposited as monolayers or incomplete monolayers. Caution must be exercised if oven curing. Exhausted, explosion-proof ovens must always be used. Deposition from aqueous solution is employed for most commercial fiberglass systems. The alkoxysilane is dissolved at 0.5–2.0% concentration in water. For less soluble silanes, 0.1% of a non-ionic surfactant is added prior to the silane and an emulsion rather than a solution is prepared. If the silane does not contain an amine group the solution is adjusted to pH 5.5 with acetic acid. The solution is either sprayed onto the substrate or employed as a bath dip. Cure is at 110–120°C for 20–30 minutes. Stability of aqueous silane solutions varies from hours for the simple alkyl silanes to weeks for the aminosilanes. Poor solubility parameters limit the use of long chain alkyl and aromatic silanes by this method. Distilled water is not necessary, but water containing fluoride ions must be avoided. Bulk deposition onto powders, e.g. filler treatment, is usually accomplished by a spray-on method. It assumes that the total amount of silane necessary is known and that sufficient adsorbed moisture is present on the filler to cause hydrolysis of the silane. The silane is prepared as a 25% solution in alcohol. The powder is placed in a high intensity solid mixer, e.g. twin cone mixer with intensifier. The solution is pumped into the agitated powder as a fine spray. In general this operation is completed within 20 minutes. Dynamic drying methods are most effective. If the filler is dried in trays, care must be taken to avoid wicking or skinning of the top layer of treated material by adjusting heat and air flow. Integral blend methods are used in composite formulations. In this method the silane is used as a simple additive. Composites can be prepared by the addition of alkoxysilanes or silazanes to dry-blends of polymer and filler prior to compounding. Generally 0.2 to 1.0 weight percent of silane (of the total mix) is dispersed by spraying the silane in an alcohol carrier onto a preblend. The addition of the silane to the non-dispersed filler is not desirable in this technique since it can lead to agglomeration. The mix is dry-blended briefly and then melt compounded. Vacuum devolatization of byproducts of silane reaction during melt compounding is necessary to achieve optimal properties. Properties are sometimes enhanced by adding 0.5–1.0% of tetrabutyl titanate or benzyldimethylamine to the silane prior to dispersal. Deposition as a primer is employed where a bulk phase is required as a transition between a substrate and a final coating. The silane is dissolved at 50% concentration in alcohol. One to three molar equivalents of water are added. The mixture is allowed to equilibrate for 15-20 mins. then diluted to 10% concentration with a higher boiling point solvent. Materials to be coated with the primer are dipped or sprayed and then cured at 110–120°C for 30–45 mins. Chlorosilanes such as SIV9110.0 may be deposited from alcohol solution. Anhydrous alcohols, particularly ethanol or isopropanol, are preferred. The chlorosilane is added to the alcohol to yield a 2–5% solution. The chlorosilane reacts with the alcohol producing an alkoxysilane and HCl. Progress of the reaction is observed by halt of HCl evolution. Mild warming of the solution (30– 40°C) promotes completion of the reaction. Part of the HCl reacts with the alcohol to produce small quantities of alkyl halide and water. The water causes formation of silanols from alkoxysilanes. The silanols condense on the substrate. Treated substrates are cured for 5–10 mins. at 110°C or allowed to stand 24 hours at room temperature. Chlorosilanes and silylamines may also be employed to treat substrates under aprotic conditions. Toluene, tetrahydrofuran, or hydrocarbon solutions are prepared containing 5% silane. The mixture is refluxed for 12–24 hours with the substrate to be treated. It is washed with the solvent. The solvent is then removed by air or explosion-proof oven drying. No further cure is necessary. This reaction involves a direct nucelophilic displacement of the silane chlorines by the surface silanol. If monolayer deposition is desired, substrates should be predried at 150°C for 4 hours. Bulk deposition results if adsorbed water is present on the substrate. This method is cumbersome for large scale preparations and rigorous controls must be established to ensure reproducible results. More reproducible coverage is obtained with monochlorosilanes. Silazanes such as SIH6110.0 and SID4612.0 may be used as treatments in concentrated form or as 10–20% solutions in aprotic solvents. In some applications parts are exposed for 5–10 minutes by dipping or in microelectronics by spin-on techniques. Optimum reactivity is as 30–50°C. An alternate method of treatment is to expose parts to 50°C vapor for 2–6 hours. Ammonia is the byproduct of silazane reaction and areas should be ventilated. REFERENCES D. Leyden, W. Collins, “Symposium and Silylated Surfaces” Gordon & Breach 1980. B. Arkles, “Tailoring Surfaces with Silanes” Chemtech 7, 766 (1977). E.P. Plueddemann, “Silane Coupling Agents” Plenum N.Y. 1982. K.L. Mittal “Silanes and Other Coupling Agents” VSP, 1992. APPENDIX Calculation of necessary silane to obtain minimum uniform multilayer coverage can be obtained knowing the values of the specific wetting surface of silane (ws) and the surface area filler amt. of silane (g) = amount of filler x surface area of filler specific wetting surface Relative surface area of common fillers m2 /g E-glass 0.1–0.12 Talc 7 Silica, ground 1–2 Si, diatomaceous 1–3.5 Kaolin 7 Calcium silicate 2.6 Clay 7 Silica, fumed 150–200 Talc 7 Si, diatomaceous 1–3.5 COUPING AGENT RECOMMENDATIONS THERMOSETS — TABLE 1 Resin Silane Class Gelest Product Code Examples diallylphthalate amine SIA0589.0 SIA0591.0 SIA0610.0 SIA059.0 styryl SIS6994.0 epoxy amine SIA0591.0 SIA0610.0 SIT8398.0 epoxy SIG5840.0 SIE4670.0 SIG5832.0 imide amine SIA0610.0 SIA0588.0 SIP6724.0 chloromethylaromatic SIC2295.5 SIC2296.2 melamine amine SIA0589.0 SIA0591.0 SIA0610.0 SIG5840.0 SIE4670.0 paralene chloromethylaromatic SIC2295.5 SIC2296.2 vinyl/allyl SIV9112.0 SIV9220.0 SIA0540.0 phenolic amine SIA0610.0 SIA0591.0 SIA0605.0 epoxy SIG5840.0 SIE4670.0 photoresist, negative silazane SID4612.0 SIH6110.0 aromatic SIP6821.0 SIP6722.6 photoresist, positive silazane SIH6110.1 phosphine SID4558.0 aromatic SIP6821.0 SIP6722.6 polyester, unsat’d amine SIA0589.0 SIA0591.0 SIA0610.0 methacrylate/vinyl SIM6487.4 SIM6487.5 SIV9112.0 styryl SIS6994.0 urethane amine SIA0591.0 SIA0610.0 SIT8398.0 epoxy SIG5840.0 SIE4670.0 SIF5832.0 isocyanate SII6455.0 THERMOPLASTICS — TABLE 2 Resin Silane Class Gelest Product Code Examples polyacetal quaternary SIS6994.0 SIT8405.0 polyacrylates methacrylate SIM6487.4 ureido / amine SIU9055.0 SIA0610.0 polyamides amine SIA0589.0 SIA0591.0 SIA0610.0 SIA0605.0 SIU9055.0 polyamide-imide amine SIA0610.0 SIA0588.0 SIP6724.0 SIC2295.5 SIC2296.2 polybutylene amine SIA0591.0 SAI0610.0 SIT8398.0 terephthalate isocyanate SII6455.0 polycarbonate amine SIA0591.0 SIA0610.0 SITU9055.0 polyethylene amine SIA0591.0 SIB1833.0 SIT8398.0 vinyl SIV9112.0 SIV9220.0 polyphenylene sulfide mercapto/thio SIM6475.0 SIB1825.0 amine SIA0591.0 SIA0610.0 SIT8398.0 polypropylene styryl SIS6994.0 aromatic SIP6821.0 SIP6722.6 SIP6724.0 polystyrene aromatic SIP6821.0 SIP6722.6 SIP6724.0 polysulfone amine SIA0591.0 SIA0610.0 SITU9055.0 polyvinylbutyral amine SIA0591.0 SIB1833.0 SIT8398.0 polyvinyl chloride amine SIA0591.0 SIB1833.0 SIT8398.0 mercapto/thio SIM6475.0 SIB1825.0 SEALANTS — TABLE 3 acrylics amine SIA0608.0 SIA0591.0 SIA0610.0 methacrylate SIM6487.4 SIM6487.5 polysulfide mercapto/thio SIM6475.0 SIB1825.0 SIT7908.0 amine SIA0591.0 SIB1833.0 SIT8398.0 RUBBERS — TABLE 4 butyl epoxy SIG5832.0 SIE4670.0 SIG5840.0 diene vinyl/cyclic olefin SIB0992.0 SIV9112.0 SIV9220.0 neoprene mercapto/thio SIM6475.0 SIB1825.0 SIT 7908.0 isoprene mercapto/thio SIM6475.0 SIB1825.0 SIT 7908.0 fluorocarbon amine SIA0610.0 SIA0588.0 SIP6724.0 epichlorhydrin amine SIA0591.0 SIB1833.0 SIT8398.0 mercapto/thio SIM6475.0 SIB1825.0 SIT7908.0 silicone allyl/vinyl SIV9112.0 SIV9220.0 SIA0540.0
Archived, Publications & Technical Reports