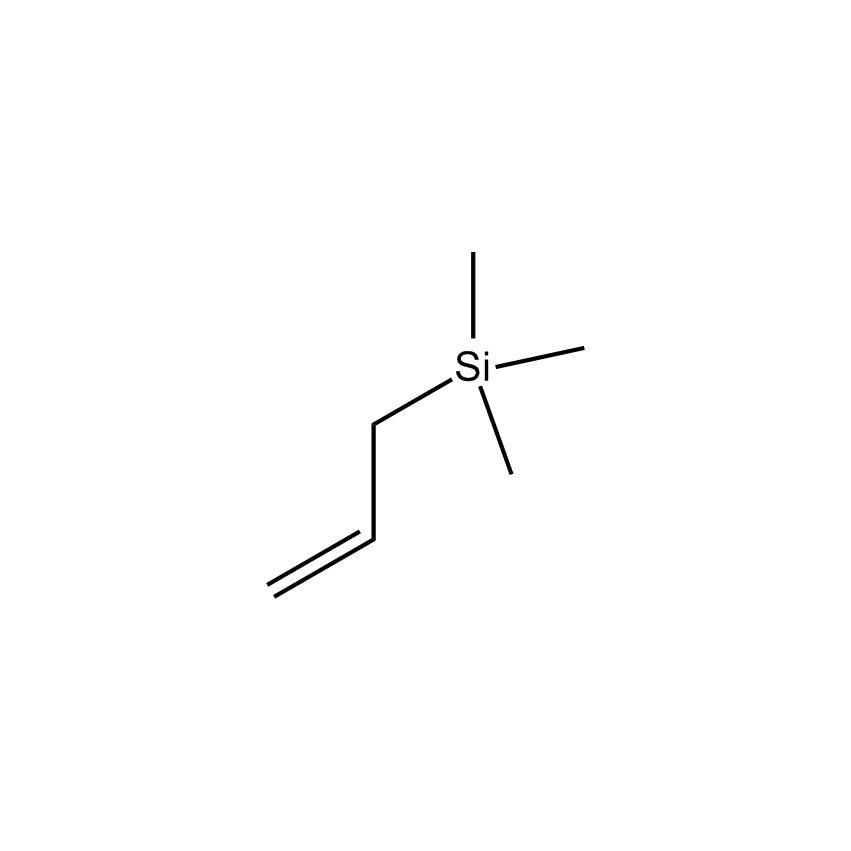
Additional Properties
- Einecs Number 212-104-5
- HMIS 2-4-0-X
- Molecular Formula C6H14Si
- Molecular Weight (g/mol) 114.26
- Purity (%) 97%
- TSCA Yes
- Boiling Point (˚C/mmHg) 85-86
- Density (g/mL) 0.7193
- Flash Point (˚C) 7 °C
- Refractive Index @ 20˚C 1.4074
Application
Key reviews.1,2,3,4
Carries out deoxygenative allylation of benzylic alcohols.10
Allylates lithium alkoxides w/ loss of the lithium oxide.11
Forms benzylic homoallylic alcohols in a 3-component reaction w/ aldehydes.12
Diallylates ketones via their ketals.13
Undergoes polymerization with zirconocene complexes.5
Provides functional termination of living carbocationic polymerized polyisobutylenes.6
Allylates dioxolanes.7
Allylates imines under fluoride catalysis.8
Provides alternative for the formation of trans-allyl-2,3-O-isopropylidene-protected pyrrolidines.9
Reference
1. Handbook for Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 14-24.
10. De, S. K.; Gibbs, R. A. Tetrahedron Lett. 2005, 46, 8345.
11. Kabalka, G. W. et al. Organometallics 2007, 26, 4112.
12. Kataki, D.; Phukan, P. Tetrahedron Lett. 2009, 50, 1958.
13. Galy, N. et al. Tetrahedron 2011, 67, 1448.
2. Weber, W. In Silicon Reagents for Organic Synthesis; Springer-Verlag: 1983, p173.
3. Fleming, I. Synthesis 1979, 761.
4. Hosomani, N. Acc. Chem. Res. 1988, 21, 200.
5. Habane, S. et al. Macromol. Chem. Phys. 1998, 199, 2211.
6. Wilczek, L. et al. J. Polym. Sci., Part A: Polym. Chem. 1987, 25, 3255.
7. Dussault, P. H.;Liu, X. Org. Lett. 1999, 1, 1391.
8. Weng, D. K. et al. J. Org. Chem. 1999, 64, 4233.
9. De Armas, P. et al. Org. Lett. 2000, 3, 3513.
Safety
Trimethylsilyl Blocking Agent
Used as a protecting group for reactive hydrogens in alcohols, amines, thiols, and carboxylic acids. Organosilanes are hydrogen-like, can be introduced in high yield, and can be removed under selective conditions. They are stable over a wide range of reaction conditions and can be removed in the presence of other functional groups, including other protecting groups. The tolerance of silylated alcohols to chemical transformations summary is presented in Table 1 of the Silicon-Based Blocking Agents brochure.
Alkenylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Allytrimethylsilane; Propenyltrimethylsilane; 3-Trimethylsilylpropene
Silicon Chemistry, Articles
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products