Additional Properties
- HMIS 2-3-0-X
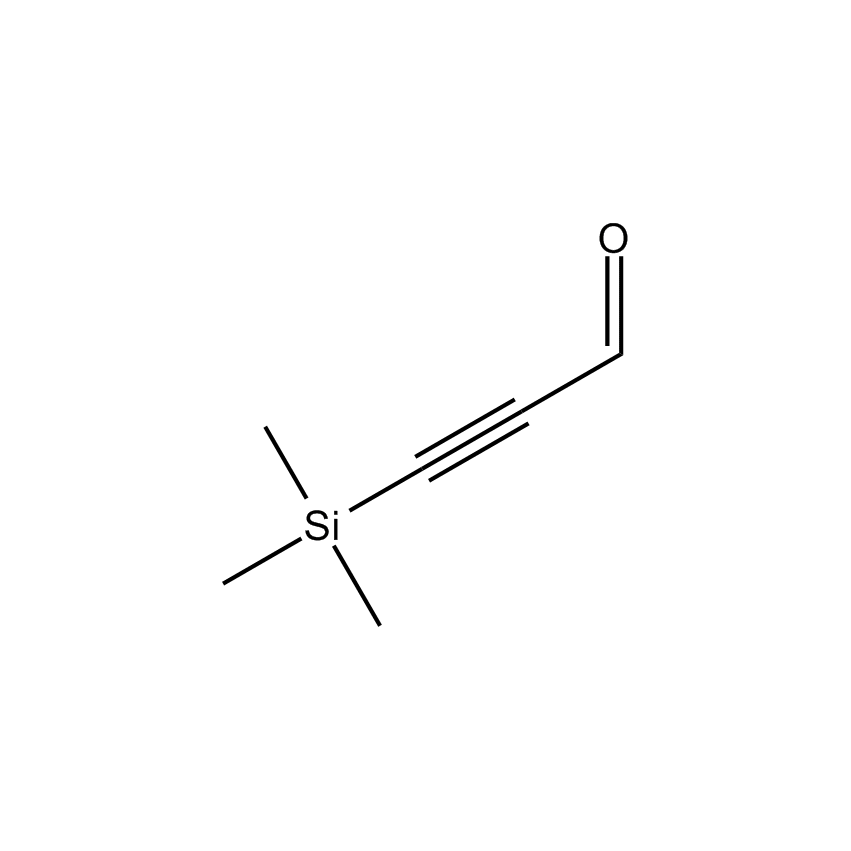
- Molecular Formula C6H10OSi
- Molecular Weight (g/mol) 126.23
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 52/30
- Density (g/mL) 0.862
- Flash Point (˚C) >25 °C
- Refractive Index @ 20˚C 1.4432
Application
Used in the preparation of 1,3,5-triynes.1
Reference
1. Mukai, C. et al. J. Org. Chem. 2001, 66, 5875.
Safety
Alkynylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
3-Trimethylsilylpropynal; 3-Trimethylsilyl-2-propyn-1-al; 3-(Trimethylsilyl)propargyl aldehyde; Trimethylsilyl)acetylenecarboxaldehyde
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products
Articles
In amongst the considerable chemistry of acetylenes there lies some unique chemistry of alkynylsilanes (silylacetylenes) some of which is reviewed herein. This unique character is exemplified not only in the silyl protection of the terminal C–H of acetylenes, but also in the ability of the silyl group to be converted into other functionalities after reaction of the alkynylsilane and to its ability to dictate and improve the regioselectivity of reactions at the triple bond. This, when combined with the possible subsequent transformations of the silyl group, makes their chemistry highly versatile and useful.