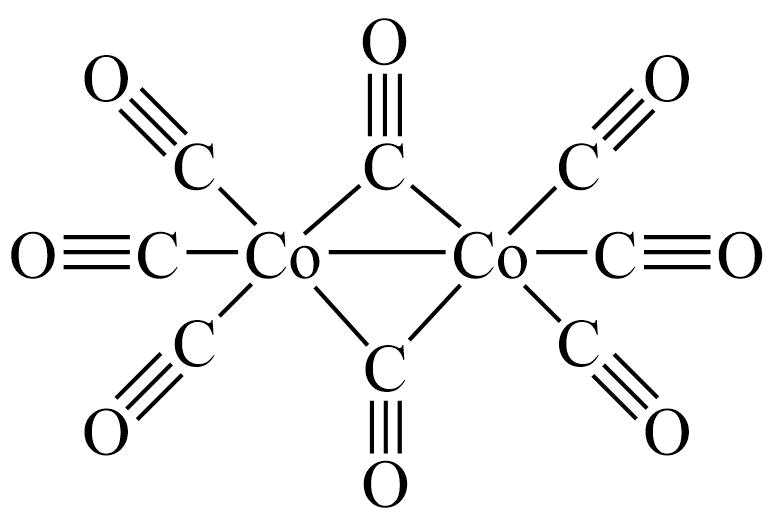
Additional Properties
- Einecs Number 233-514-0
- Synonyms DICOBALT OCTACARBONYL
- HMIS 3-3-1-X
- Molecular Formula C8Co2O8
- Molecular Weight (g/mol) 341.95
- TSCA Yes
- Boiling Point (˚C/mmHg) NA
- Density (g/mL) 1.73
- Melting Point (˚C) 51-2° dec
Application
In combination with SiH4 forms CoSi by CVD.1
Catalyst for conversion of olefins, alkynes, and CO to cyclopentenones.2
Reference
1. West, G. et al. Appl. Phys. Lett. 1988, 53, 740.
2. Comprehensive Organic Synthesis, Vol. 5 9.1, 1037, 1991
Safety
ALD Material
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
CVD Material
The growth of thin films via chemical vapor deposition (CVD) is an industrially significant process with a wide array of applications, notably in microelectronic device fabrication. A volatilized precursor (such as a silane, organometallic or metal coordination complex) is passed over a heated substrate. Thermal decomposition of the precursor produces a thin-film deposit, and ideally, the ligands associated with the precursor are cleanly lost to the gas phase as reaction products. Compared to other thin-film production techniques, CVD offers several significant advantages, most notably the potential for effecting selective deposition and lower processing temperatures. Many metal CVD depositions are autocatalytic. Growth of such thin films is characterized by an induction period, which is a consequence of the higher barriers that relate to the activation of the precursor on a non-native substrate. CVD is the preferred deposition method for fabricating optical storage, as it is a well-established method with good scalability, reproducibility, and uniformity. It is also capable of high rates and good composition control.
Cobalt carbonyl; Octacarbonyldicobalt; Dicobalt octacarbonyl; Cobalt tetracarbonyl dimer
Metal-Organic Chemistry, Applied Chemistry & Physics
Metal-Organic Chemistry, Applied Chemistry & Physics
Metal-Organic Chemistry
Cobalt metallic films are the subject of an ever-expanding academic and industrial interest for incorporation into a multitude of new technological applications. This report reviews the state-of-the art chemistry and deposition techniques for cobalt thin films, highlighting innovations in cobalt metal-organic chemical vapor deposition (MOCVD), plasma and thermal atomic layer deposition (ALD), as well as pulsed MOCVD technologies, and focusing on cobalt source precursors, thin and ultrathin film growth processes, and the resulting effects on film composition, resistivity and other pertinent properties.
Silicon Chemistry, Applied Technology, Metal-Organic Chemistry, Applied Chemistry & Physics