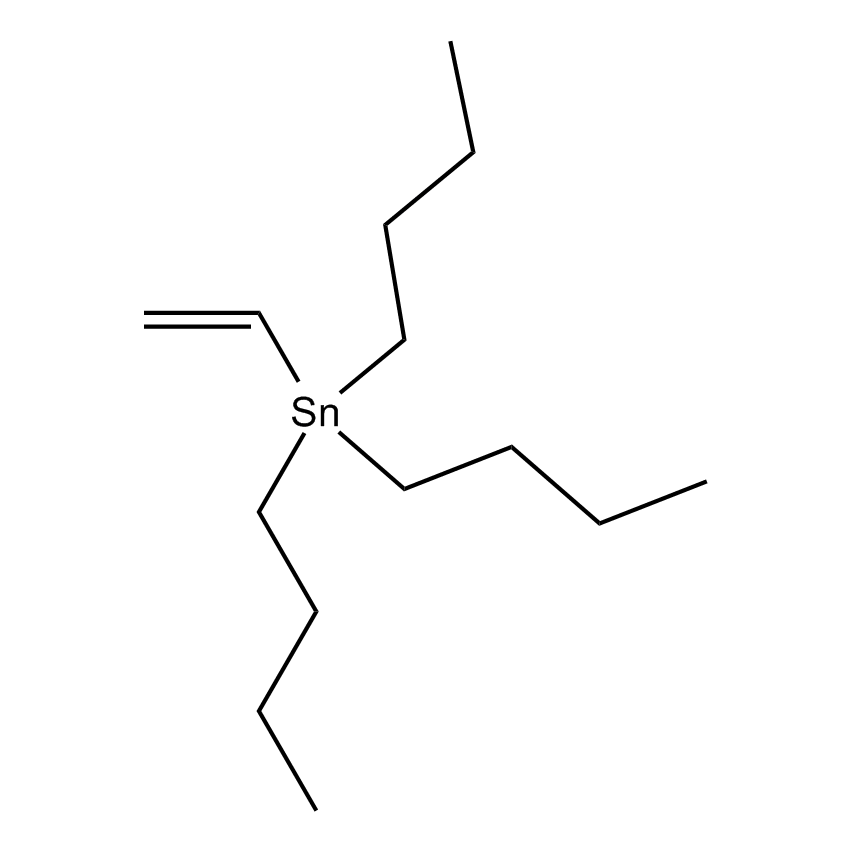
Additional Properties
- Einecs Number 231-291-4
- HMIS 2-2-0-X
- Molecular Formula C14H30Sn
- Molecular Weight (g/mol) 317.08
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 104-106/3.5
- Density (g/mL) 1.085
- Flash Point (˚C) >110 °C
- Refractive Index @ 20˚C 1.4776
Application
Reviewed.1
Intermediate for vinyllithium by exchange with phenyllithium.2
With CO forms divinyl ketones on reaction with vinyl iodides.3
Reacts with IC6H4CHO in DMF with PdCl2(CH3CN)2 to yield CH2=CHC6H4CHO.4
Reacts with halopyrimidines to yield vinylpyrimidines.5
Employed in one-pot synthesis of pyrroles.6
Reference
1. Scott, W. J.; Moretto, A. F. in Encyclopedia of Reagents for Organic Synthesis 1995, Volume 8, 5503-5506.
2. Seyferth, D. et al. J. Am. Chem. Soc. 1961, 83, 3583.
3. Goure, W. et al. F&F: Vol. 12, p 45; J. Am. Chem. Soc. 1984, 106, 6417.
4. Bumagin, N. Chem. Abstr. 111, 214179; Metal Org. Chem. 1989, 2, 475.
5. Kondo, Y. et al. Chem. Pharm. Bull. 1989, 37, 2814.
6. Nakamura, M. et al. Org. Lett. 1999, 1, 1505.
Safety
Organotin Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Vinyltri-n-butyltin; Tributylethenylstannane; Tributylvinylstannane
Metal-Organic Chemistry, Articles
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products