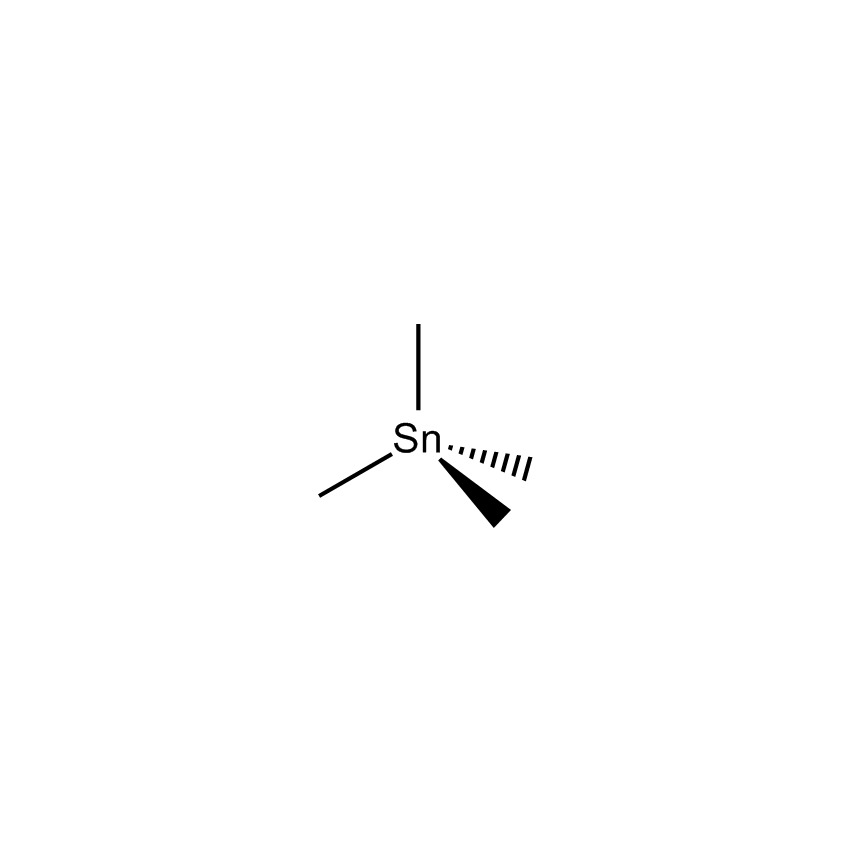
Additional Properties
- Einecs Number 209-833-6
- HMIS 3-4-0-X
- Molecular Formula C4H12Sn
- Molecular Weight (g/mol) 178.83
- Purity (%) 97%
- TSCA Yes
- Delta H Vaporization (kJ/mol) 6.8 kcal/mole
- Boiling Point (˚C/mmHg) 74-75
- Density (g/mL) 1.291
- Flash Point (˚C) -12 °C
- Melting Point (˚C) -54°
- Refractive Index @ 20˚C 1.4410
Application
Reviewed.1
Forms transparent conductive oxides for photovoltaics by PECVD.10
Safety and handling considerations.11
In combination with WCl6 catalyzes olefin metathesis.2
Allows synthesis of even numbered alkanes.3
Converts acid chlorides to methyl ketones with benzylchlorobis(triphenyl phosphine)palladium.4,5
Forms aryl methyl ketones from aryl halides and CO in the presence of dicarbonylbis(triphenylphosphine)nickel.6
For CVD of tin oxide transparent conductive electrodes on glass for photovoltaics and sensors.7
Pyrolyzed in vacuum to tin at 600-750°.8
Pyrolyzed oxidatively to SnO at 350-600°.9
Reference
1. Scott, W. J.; Jones, J. H.; Moretto, A. F. in Encyclopedia of Reagents for Organic Synthesis 1995, Volume 8, 4823-4825.
10. Chandra, H. et al. Am. Vac. Soc. 2008, 55, TFThA10.
11. Kalb, P. et al. Report BNL-52123, 1987 Order NTIS #DE89005936.
2. van Dam, P. et al. J. Chem. Soc., Chem. Commun. 1972, 1221.
3. Gibson, T. et al. J. Org. Chem. 1981, 46, 1821.
4. Milstein, D. et al. J. Org. Chem. 1979, 44, 1613.
5. Labadie, J. et al. J. Am. Chem. Soc. 1983, 105, 6129.
6. Tanaka, M. Synthesis 1981, 47.
7. Inagaki, N. J. Appl. Polym. Sci. 1989, 37, 2341.
8. Svoboda, G. et al. Ind. Eng. Chem. Res. 1992, 31, 19.
9. Borman, C. et al. J. Electrochem. Soc. 1989, 136, 3820.
Safety
ALD Material
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
Tetramethyltin; Tetramethylstannane