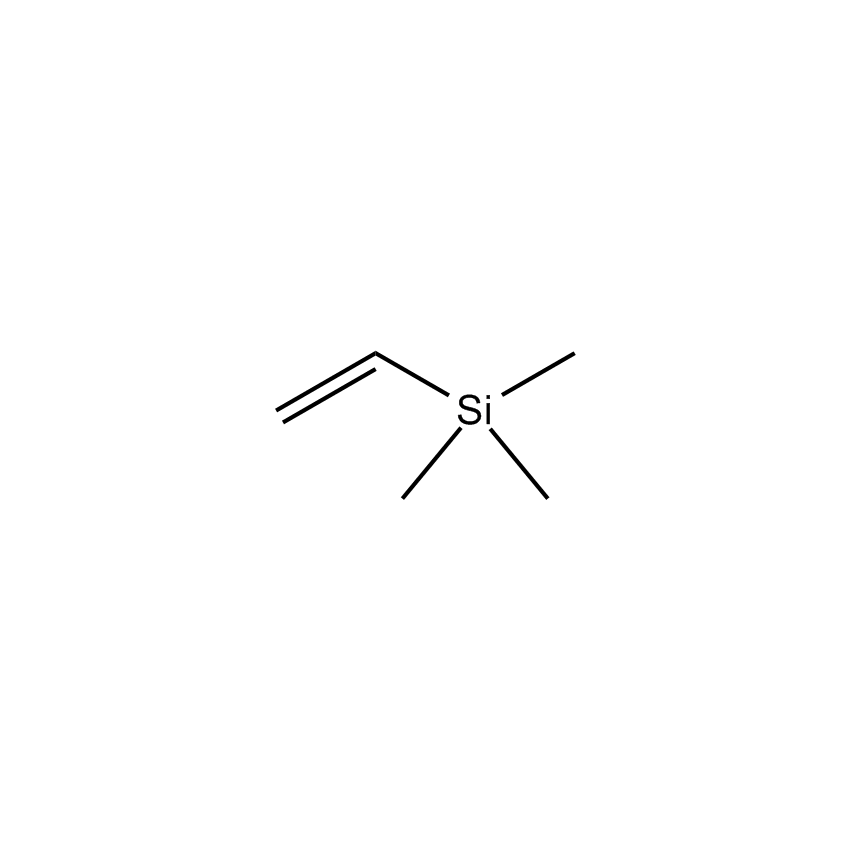
Additional Properties
- Einecs Number 212-042-9
- HMIS 2-4-0-X
- Molecular Formula C5H12Si
- Molecular Weight (g/mol) 100.24
- Purity (%) 97%
- TSCA Yes
- Autoignition Temp (˚C) 295
- Boiling Point (˚C/mmHg) 55
- Density (g/mL) 0.6903
- Flash Point (˚C) -20 °C
- Melting Point (˚C) -132°
- Refractive Index @ 20˚C 1.3910
- Additional Viscosity (cSt) '0.5, 20
- Viscosity at 25 ˚C (cSt) 0.5
Application
Review of synthetic utility.1
Polymerization catalyzed by alkyllithium compounds.2,3,4
Synthetic reactions of vinylsilanes reviewed.5,6
Undergoes Heck coupling to (E)-?-substituted vinyltrimethylsilanes, which can be cross-coupled further.7
Reacts w/ azides to form trimethylsilyl-substituted aziridines.8
Fieser
F&F: Vol. 9, p 498; Vol. 10, p 44; Vol. 11, p 41; Vol. 12, p 566.
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 755-761.
2. Oku, J. et al. Polymer J. 1991, 23, 1377; Macromolecules 1992, 25, 2780.
3. Gan, Y. et al. Macromolecules 1996, 29, 8285.
4. Rickle, G. J. Macromol. Sci. 1987, A24, 93.
5. Hudrlik, P. In New Applications of Organometallic Reagents in Organic Synthesis; Seyferth, D., Ed.; Elsevier: 1976.
6. F&F: Vol. 5, p 375; Vol. 6, p 637.
7. Jeffery, T. Tetrahedron Lett. 1999, 40, 1673.
8. Bassindale, A. R. et al. J. Chem. Soc., Perkin Trans. 1 2000, 1173.
Safety
Alkenylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Vinyltrimethylsilane; Ethenyltrimethylsilane; Trimethylsilylethene; Trimethylvinylsilane
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products