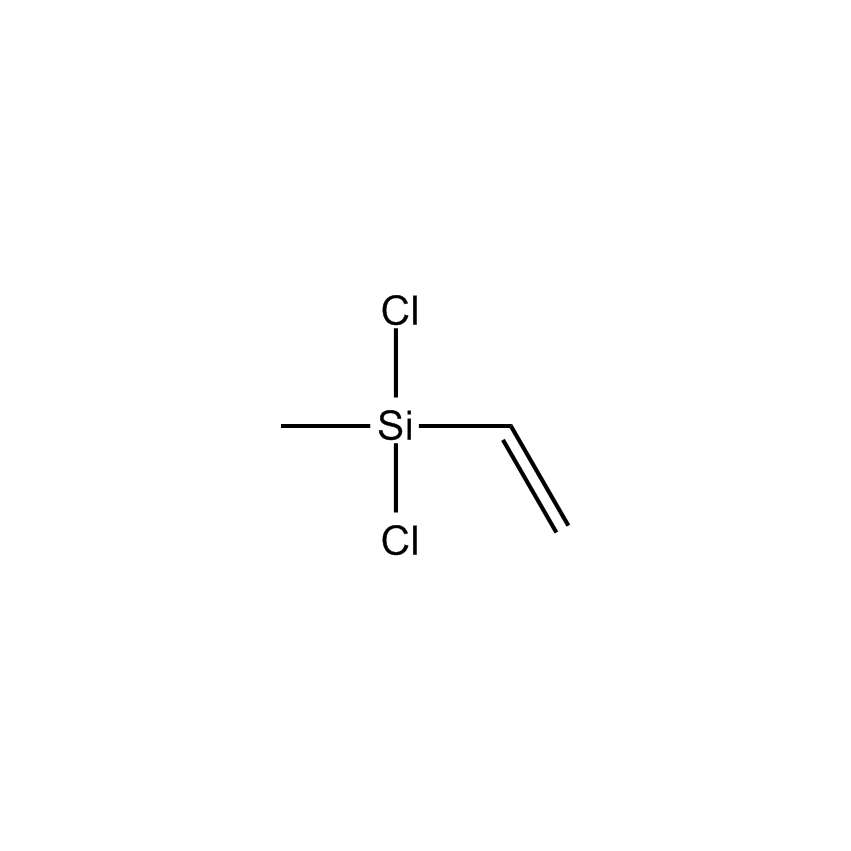
Additional Properties
- Einecs Number 204-710-3
- HMIS 3-4-1-X
- Molecular Formula C3H6Cl2Si
- Molecular Weight (g/mol) 141.07
- Purity (%) 97%
- TSCA Yes
- Delta H Vaporization (kJ/mol) 8.1 kcal/mole
- Boiling Point (˚C/mmHg) 92-93
- Density (g/mL) 1.087
- Flash Point (˚C) 4 °C
- Melting Point (˚C) -78°
- Refractive Index @ 20˚C 1.4270
- Viscosity at 25 ˚C (cSt) 0.7
Application
Used as a tether in synthesis of C-glycosides.1
Reacts to vinylate aryl halides under NaOH-moderated conditions.2
Reference
1. Shuto, S. et al. J. Org. Chem. 2000, 65, 5547.
2. Hagiwara, E. et al. Tetrahedron Lett. 1997, 38, 439.
Safety
Alkenylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Vinylmethyldichlorosilane; Dichlorovinylmethylsilane; Methylvinyldichlorosilane; Dichloroethenylmethylsilane
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products