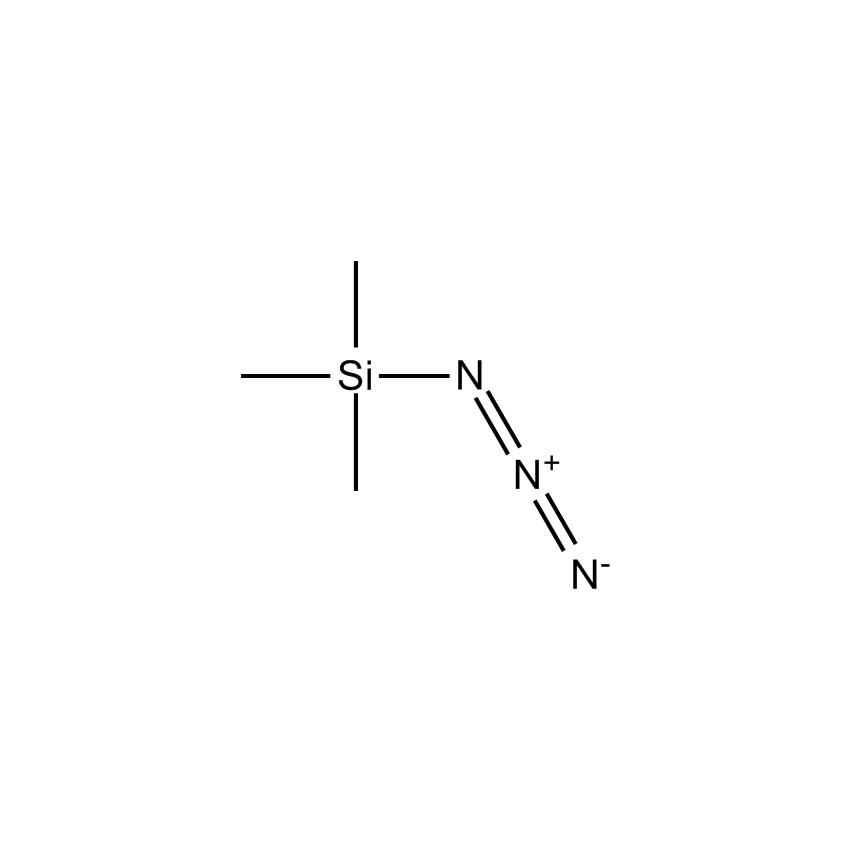Additional Properties
- Einecs Number 225-078-5
- Synonyms AZIDOTRIMETHYLSILANE
- HMIS 4-3-2-X
- Molecular Formula C3H9N3Si
- Molecular Weight (g/mol) 115.21
- Purity (%) 96%
- TSCA Yes
- Autoignition Temp (˚C) 300
- Boiling Point (˚C/mmHg) 95-96
- Density (g/mL) 0.876
- Flash Point (˚C) 30 °C
- Melting Point (˚C) -95°
- Refractive Index @ 20˚C 1.4140
Application
Review of synthetic utility.1
Used in the preparation of glycosyl azides.2
Reacts with terminal acetylenes and allyl carbonates to give 1-allyl-3-substituted 1,2,3-triazoles.3
Used to prepare the energetically unstable 2,5,8-triazido-s-heptazine.4
Converts alcohols directly to azides with complete inversion.5
Used in a one-pot, click-conversion of amines to triazoles.6
Converts allyl alcohols to allyl azides.7
Reaction with bridged bicyclic olefins leads to 1,2,3-triazoles by retro-Diels-Alder reaction.8
Fieser
F&F: Vol. 1, p 1236; Vol. 3, p 316; Vol. 5, p 354, p 719; Vol. 6, p 561, p 632; Vol. 9, p 21; Vol. 10, p 14, p 113; Vol. 11, p 32; Vol. 12, p 37; Vol. 13, p 34; Vol. 14, p 25; Vol. 15, p 16, p 342; Vol. 16, p 17; Vol. 17, p 23,p 157,p 378; Vol. 18, p 379, p 380; Vol. 19, p 371; Vol. 20, p 201,p 403; Vol. 21, p 151.
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 26-31.
2. Soli, E. D.; DeShong, P. J. Org. Chem. 1999, 64, 9724.
3. Kamijo, S. et al. J. Am. Chem. Soc. 2003, 125, 7786.
4. Miller, D. R. et al. J. Am. Chem. Soc. 2004, 126, 5372.
5. Mukaiyama, T. et al. Chem. Lett. 2008, 37, 1072.
6. Savonnet, M. et al. J. Am. Chem. Soc. 2010, 132, 4418.
7. Ruepling, M. et al. Org. Lett. 2012, 15, 768.
8. Peterson, W. et al. J. Organomet. Chem. 1976, 121, 285.
Safety
Archived, Publications & Technical Reports
Applied Technology
This overview provides an introduction and comparison of emerging processing technologies that represent the best contenders to satisfy future demands for ultrathin film applications.