Additional Properties
- HMIS 3-2-1-X
- Molecular Formula C16H37NO5Si2
- Molecular Weight (g/mol) 379.64
- TSCA Yes (L)Low Volume Exemption
- Boiling Point (˚C/mmHg) 136-138/1
- Density (g/mL) 0.974
- Refractive Index @ 20˚C 1.4322
Safety
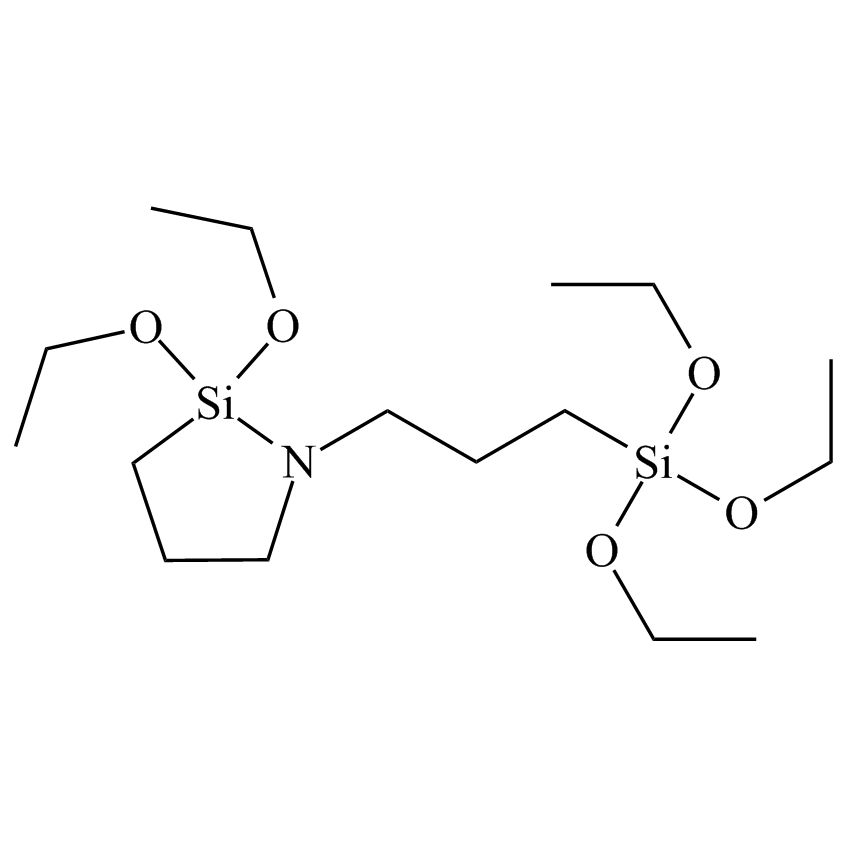
1-(3-triethoxysilyl)propyl-2,2-diethoxy-1-aza-2-silayclopentane
Cross-Linking Cyclic Azasilane
Cyclic azasilanes react rapidly, less than one minute, with any and all surface hydroxyl groups is therefore of unique interest for surface modification. Volatile cyclic azasilanes afford high functional density monolayers on inorganic surfaces such as nanoparticles and other nanofeatured substrates without a hydrolysis step or the formation of byproducts. They exploit the Si–N and Si–O bond energy differences affording a thermodynamically favorable ring-opening reaction with surface hydroxyls at ambient temperature.
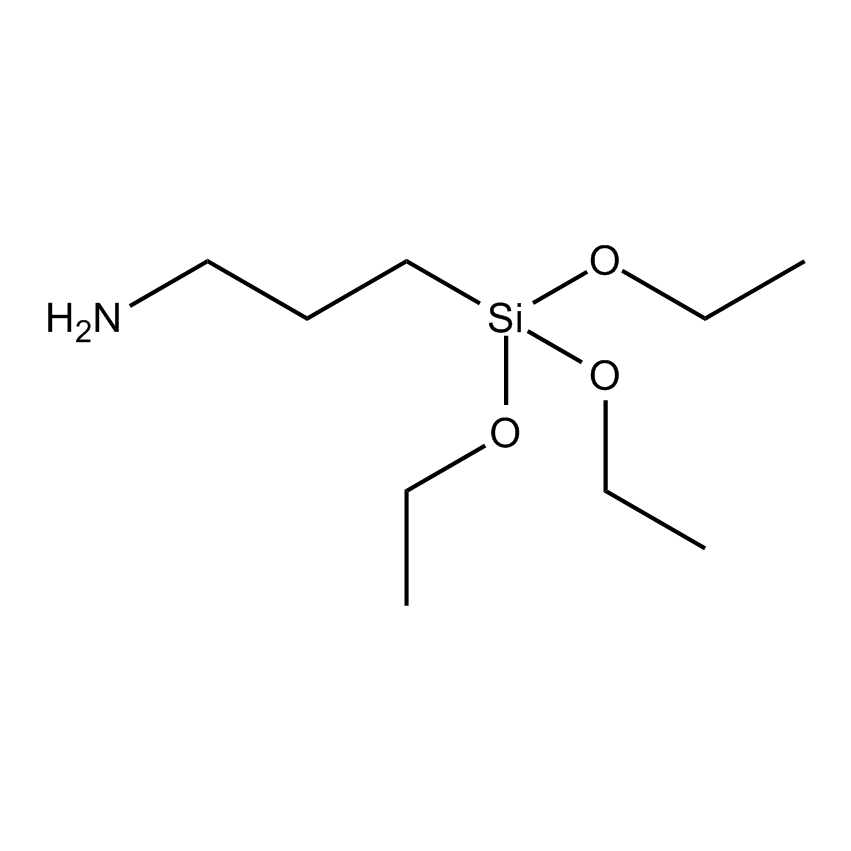
Amine Functional Silane Coupling Agent
Silane coupling agents have the ability to form a durable bond between organic and inorganic materials to generate desired heterogeneous environments or to incorporate the bulk properties of different phases into a uniform composite structure. The general formula has two classes of functionality. The hydrolyzable group forms stable condensation products with siliceous surfaces and other oxides such as those of aluminum, zirconium, tin, titanium, and nickel. The organofunctional group alters the wetting or adhesion characteristics of the substrate, utilizes the substrate to catalyze chemical transformations at the heterogeneous interface, orders the interfacial region, or modifies its partition characteristics, and significantly effects the covalent bond between organic and inorganic materials.
Dipodal Silane
Dipodal silanes are a series of adhesion promoters that have intrinsic hydrolytic stabilities up to ~10,000 times greater than conventional silanes and are used in applications such as plastic optics, multilayer printed circuit boards and as adhesive primers for ferrous and nonferrous metals. They have the ability to form up to six bonds to a substrate compared to conventional silanes with the ability to form only three bonds to a substrate. Many conventional coupling agents are frequently used in combination with 10-40% of a non-functional dipodal silane, where the conventional coupling agent provides the appropriate functionality for the application, and the non-functional dipodal silane provides increased durability. Also known as bis-silanes additives enhance hydrolytic stability, which impacts on increased product shelf life, ensures better substrate bonding and also leads to improved mechanical properties in coatings as well as composite applications.
Applied Technology
Silicon Chemistry, Applied Technology
How cyclic azasilanes and cyclic thiasilanes, a new class of silane coupling agents, modify nanoparticles and create nano-scale features.
Silicon Chemistry, Applied Technology
Silicon Chemistry, Articles
Manufacturing growth opportunities exist in the United States for alternative energy (photovoltaic modules, fuel cells, wind turbines) and medical and healthcare (surgical devices, dental implants and drug delivery) applications. More traditional markets where growth is expected include automotive, construction and specialty packaging.
Silicon Chemistry, Applied Technology
Applied Technology
This overview provides an introduction and comparison of emerging processing technologies that represent the best contenders to satisfy future demands for ultrathin film applications.
Silicon Chemistry, Applied Technology
Applied Technology
Silicon Chemistry, Applied Technology
Thiasilacyclopentane (TSCP) and azasilacyclopentane (ASCP) heteroatom cyclics reagents can be extended to “simultaneous doubleclicking” when both inorganic and organic substrates are present at the onset of the reaction. The simultaneous double-click depends on a first ring-opening click with an inorganic substrate that is complete in ~1 s at 30 °C and results in the reveal of a cryptic mercaptan or secondary amine group, which can then participate in a second click with an organic substrate.
Applied Technology