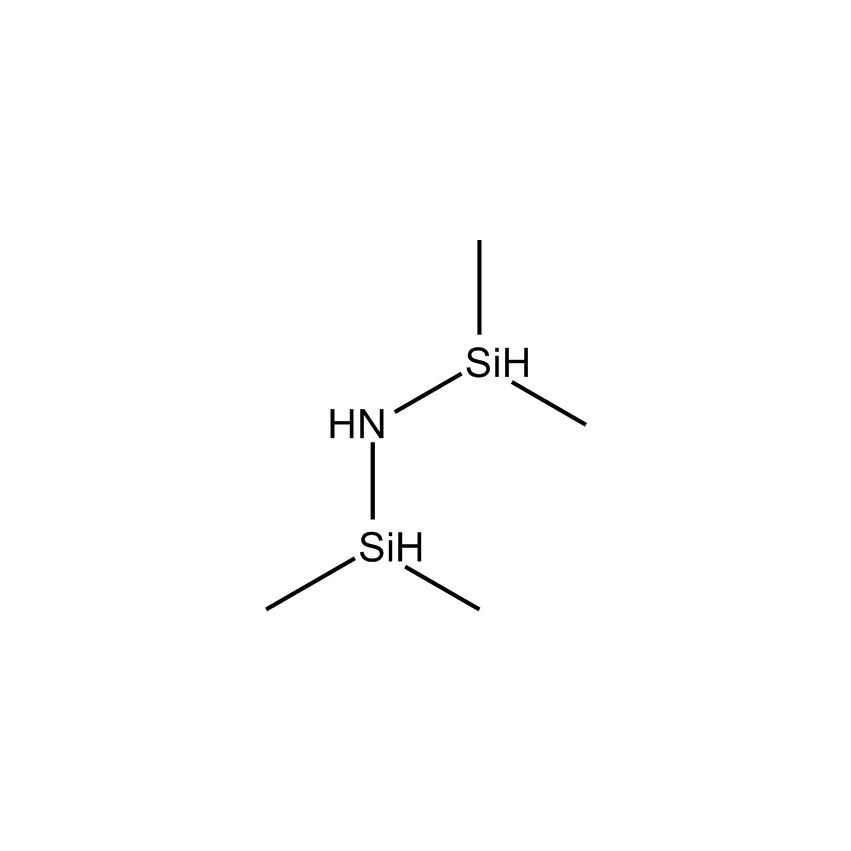
Additional Properties
- Einecs Number 240-072-2
- HMIS 3-4-1-X
- Molecular Formula C4H15NSi2
- Molecular Weight (g/mol) 133.34
- Purity (%) 97%
- TSCA Yes
- Autoignition Temp (˚C) 208
- Boiling Point (˚C/mmHg) 99-100
- Density (g/mL) 0.766
- Flash Point (˚C) 11 °C
- Refractive Index @ 20˚C 1.4044
Application
Review of synthetic utility.1
Converts unsaturated alcohols to diols.2
Converts homopropargylic alcohols to ?-hydroxy ketones in a three-step process.3
Employed in the silylation/hydrosilylation of allylic alcohols leading to useful cyclic alkoxysilane intermediates.4
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 488-490.
2. Tamao, K. et al. J. Am. Chem. Soc. 1988, 110, 3712.
3. Marshall, J. A.; Yanik, M.W. Org. Lett. 2000, 2, 2173.
4. Tamao, K. et al. J. Am. Chem. Soc. 1986, 108, 6090.
Safety
ALD Material
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
1,1,3,3-Tetramethyldisilazane; Bis(dimethylsilyl)amine; TMDS; TMDZ
Silicon Chemistry, Applied Technology
This article provides an overview of the state-of-the-art chemistry and processing technologies for silicon nitride and silicon nitride- rich films, i.e., silicon nitride with C inclusion, both in hydrogenated (SiNx:H and SiNx:H(C)) and non-hydrogenated (SiNx and SiNx(C)) forms. The emphasis is on emerging trends and innovations in these SiNx material system technologies, with focus on Si and N source chemistries and thin film growth processes, including their primary effects on resulting film properties. It also illustrates that SiNx and its SiNx(C) derivative are the focus of an ever-growing research and manufacturing interest and that their potential usages are expanding into new technological areas.