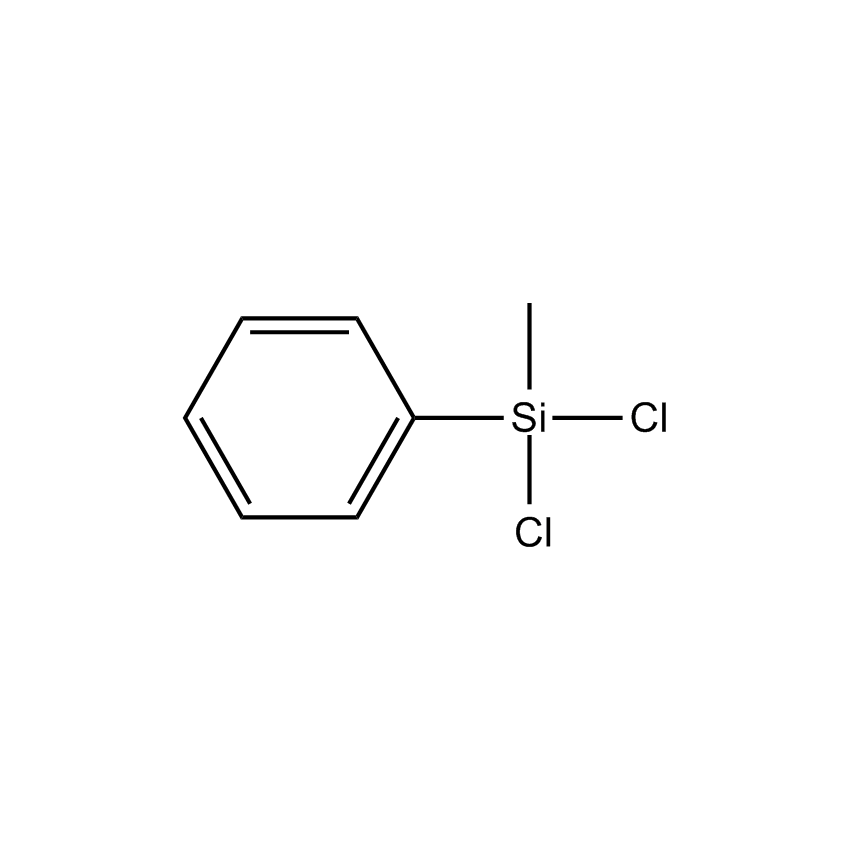
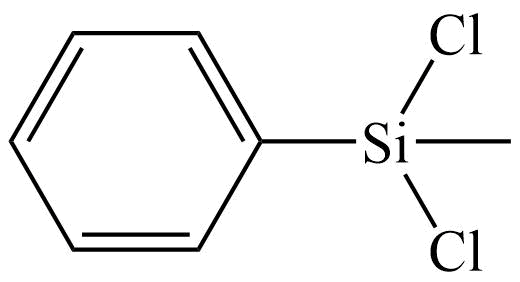
Additional Properties
- Einecs Number 205-746-2
- HMIS 3-2-1-X
- Molecular Formula C7H8Cl2Si
- Molecular Weight (g/mol) 191.13
- Purity (%) 97%
- TSCA Yes
- Delta H Vaporization (kJ/mol) 11.5 kcal/mole
- Autoignition Temp (˚C) >400
- Boiling Point (˚C/mmHg) 205-206
- Density (g/mL) 1.187
- Flash Point (˚C) 82 °C
- Melting Point (˚C) -53°
- Refractive Index @ 20˚C 1.5180
- Additional Viscosity (cSt) '1.2, 20
- Viscosity at 25 ˚C (cSt) 1.2
Fieser
F&F: Vol. 10, p 91; Vol. 11, p 247; Vol. 12, p 231.
Safety
Aromatic Silane - Conventional Surface Bonding
Aliphatic, fluorinated aliphatic or substituted aromatic hydrocarbon substituents are the hydrophobic entities which enable silanes to induce surface hydrophobicity. The organic substitution of the silane must be non-polar. The hydrophobic effect of the organic substitution can be related to the free energy of transfer of hydrocarbon molecules from an aqueous phase to a homogeneous hydrocarbon phase. A successful hydrophobic coating must eliminate or mitigate hydrogen bonding and shield polar surfaces from interaction with water by creating a non-polar interphase. Although silane and silicone derived coatings are in general the most hydrophobic, they maintain a high degree of permeability to water vapor. This allows coatings to breathe and reduce deterioration at the coating interface associated with entrapped water. Since ions are not transported through non-polar silane and silicone coatings, they offer protection to composite structures ranging from pigmented coatings to rebar reinforced concrete. A selection guide for hydrophobic silanes can be found on pages 22-31 of the Hydrophobicity, Hydrophilicity and Silane Surface Modification brochure.
Arylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Phenylmethyldichlorosilane; Methylphenyldichlorosilane; Dichloromethylphenylsilane
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products