Additional Properties
- Einecs Number 257-747-2
- HMIS 3-2-1-X
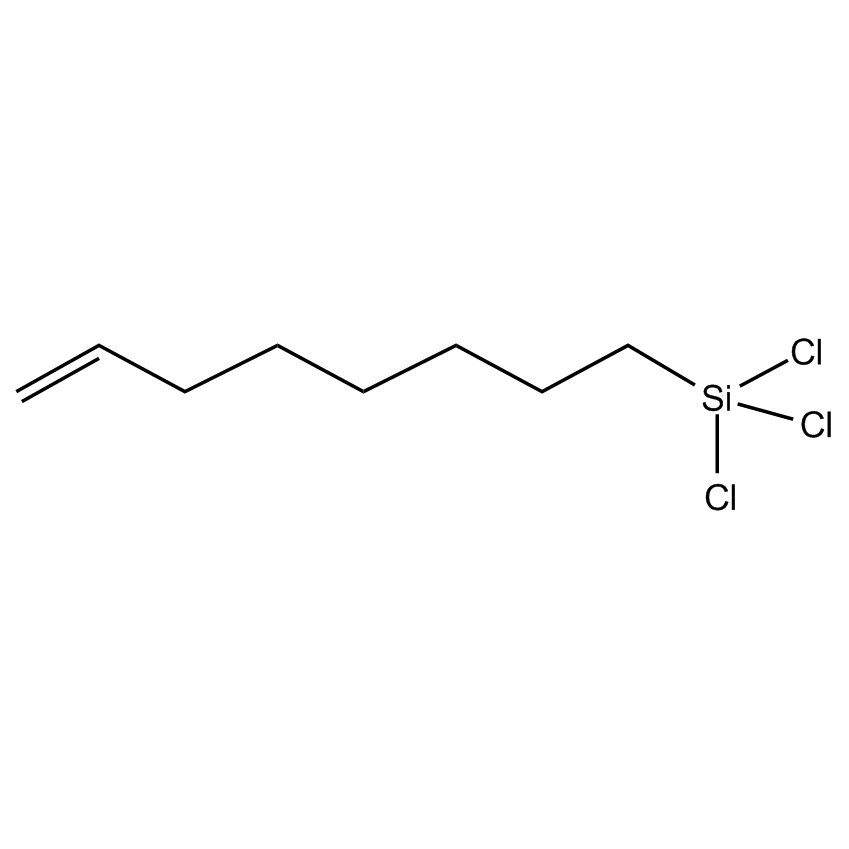
- Molecular Formula C8H15Cl3Si
- Molecular Weight (g/mol) 245.65
- Purity (%) 97%
- TSCA Yes
- Boiling Point (˚C/mmHg) 223-224
- Density (g/mL) 1.07
- Flash Point (˚C) 94 °C
- Refractive Index @ 20˚C 1.4578
Application
Immobilizes DNA at terminus.1
Reference
1. Bensimon, A. et al. Science 1994, 265, 2096.
Safety
ALD Materials
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
Olefin Functional Silane Coupling Agent
Silane coupling agents have the ability to form a durable bond between organic and inorganic materials to generate desired heterogeneous environments or to incorporate the bulk properties of different phases into a uniform composite structure. The general formula has two classes of functionality. The hydrolyzable group forms stable condensation products with siliceous surfaces and other oxides such as those of aluminum, zirconium, tin, titanium, and nickel. The organofunctional group alters the wetting or adhesion characteristics of the substrate, utilizes the substrate to catalyze chemical transformations at the heterogeneous interface, orders the interfacial region, or modifies its partition characteristics, and significantly effects the covalent bond between organic and inorganic materials.
7-Octenyltrichlorosilane; Trichlorosilyloctene; Trichloro-7-octenylsilane