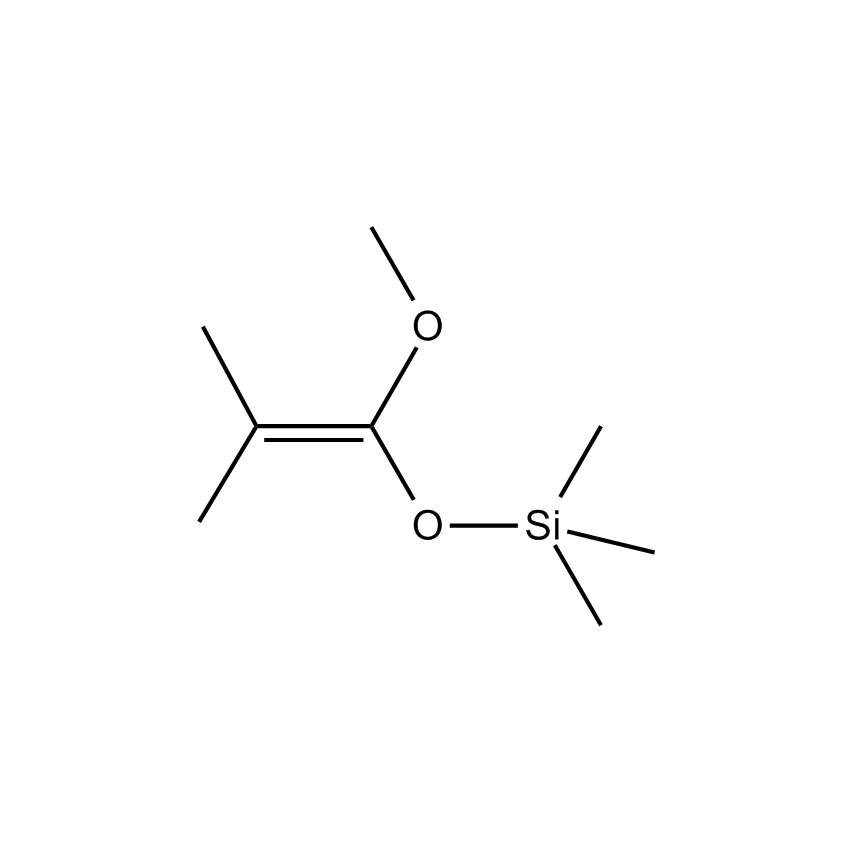
Additional Properties
- Synonyms METHYL(TRIMETHYLSILYL)DIMETHYLKETENE ACETAL
- HMIS 2-2-1-X
- Molecular Formula C8H18O2Si
- Molecular Weight (g/mol) 174.31
- Purity (%) 97%
- TSCA Yes
- Boiling Point (˚C/mmHg) 57/15
- Density (g/mL) 0.858
- Flash Point (˚C) 43 °C
- Refractive Index @ 20˚C 1.4150
Application
Review of synthetic utility.1
Initiator for group transfer polymerization.2
With SmI2 converts ketones and aldehydes to silyl enol ethers.3
Cross-couples with aryl bromides.4
Undergoes ?-arylation to form 2-aryl acetates.5
Reacts with fullerenes to form ?-fullerenyl esters.6
Adds to ethyl propiolate in a 2+2 cycloaddition fashion.7
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 376-380.
2. Webster, O. et al. Science 1983, 222, 39.
3. Hydrio, J. et al. Synthesis 1997, 68, 1.
4. Hama, T. et al. J. Am. Chem. Soc. 2003, 125, 11176.
5. Liu, X.; Hartwig, J. F. J. Am. Chem. Soc. 2004, 126, 5182.
6. Nakamura, E. et al. Org. Lett. 2008, 10, 4923.
7. Quendo, A.; Rousseau, Tetrahedron Lett. 1988, 29, 6443.
Safety
Trimethylsilyl Blocking Agent
Used as a protecting group for reactive hydrogens in alcohols, amines, thiols, and carboxylic acids. Organosilanes are hydrogen-like, can be introduced in high yield, and can be removed under selective conditions. They are stable over a wide range of reaction conditions and can be removed in the presence of other functional groups, including other protecting groups. The tolerance of silylated alcohols to chemical transformations summary is presented in Table 1 of the Silicon-Based Blocking Agents brochure.
1- Methoxy-1-trimethysiloxy-2-methyl-1-propene; Methyl(trimethylsilyl)dimethylketene acetal; 1-Methoxy-2-methyl-1-(trimethylsiloxy)propene