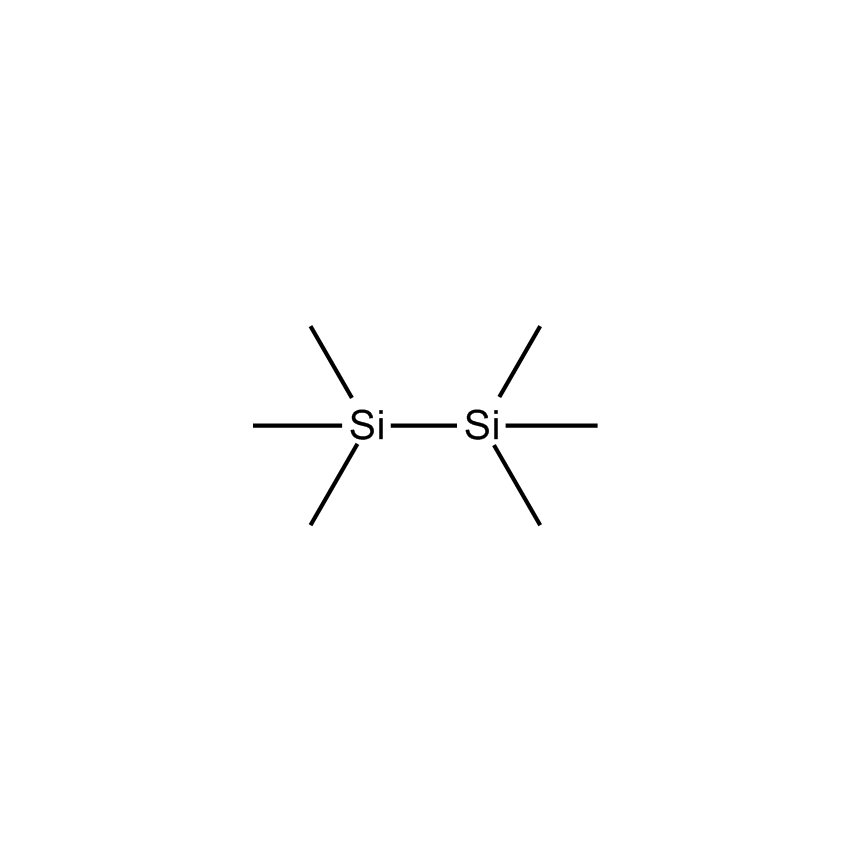Additional Properties
- Einecs Number 215-911-0
- Synonyms HMD
- HMIS 2-4-0-X
- Molecular Formula C6H18Si2
- Molecular Weight (g/mol) 146.38
- TSCA Yes
- Delta H Vaporization (kJ/mol) 9.5 kcal/mole
- Boiling Point (˚C/mmHg) 112-113
- Density (g/mL) 0.7293
- Flash Point (˚C) -1 °C
- Melting Point (˚C) 12-14°
- Refractive Index @ 20˚C 1.4214
- Viscosity at 25 ˚C (cSt) '1
Application
Review of synthetic utility.1
Source for trimethylsilyl anion.2,3
Replaces aromatic nitriles with TMS groups in presence of [RhCl(cod)]2.4
Precursor for CVD of silicon carbide.5
Brings about the homocoupling of arenesulfonyl chlorides in the presence of Pd2(dba)3.6
Used as a solvent for the direct borylation of fluoroaromatics.7
Reacts with alkynes to form siloles.8
Undergoes the silylation of acid chlorides to give acylsilanes.9
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 278-284.
2. Shippey, M. A. et al. J. Org. Chem. 1977, 42, 2654.
3. F&F: Vol. 10, p 96; Vol.11, p 253.
4. Tobisu, M. et al. J. Am. Chem. Soc. 2006, 128, 4152.
5. Thin Solid Films 1999, 252, 13.
6. Kashiwabara, T.; Tanaka, M. Tetrahedron Lett. 2005, 46, 7125.
7. Teltewskoi, M. et al. Angew. Chem., Int. Ed. Engl. 2010, 49, 3947.
8. Akagawa, K. et al. Synlett 2011, 22, 813.
9. Capperucci, A. et al. J. Org. Chem. 1988, 53, 3612.
Safety
Hexamethyldisilane; HMD; 2,2,3,3-Tetramethyl-2,3-disilabutane
Silicon Chemistry, Applied Technology