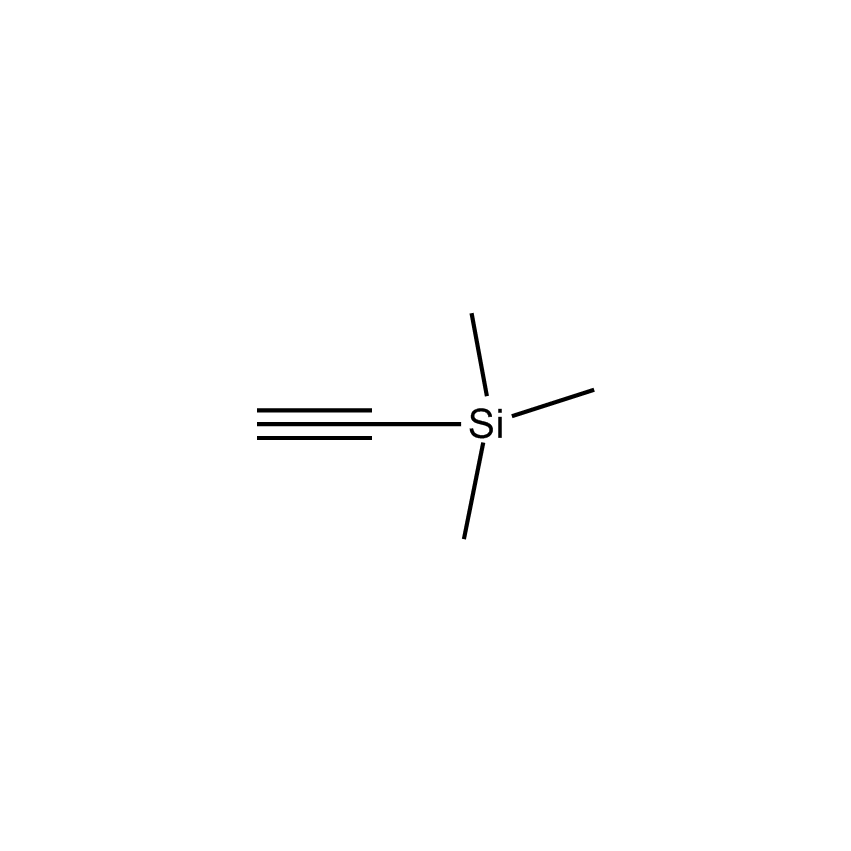Additional Properties
- Einecs Number 213-919-9
- Synonyms TRIMETHYLSILYLACETYLENE
- HMIS 2-4-1-X
- Molecular Formula C5H10Si
- Molecular Weight (g/mol) 98.22
- Purity (%) 97%
- TSCA Yes
- Autoignition Temp (˚C) 310
- Boiling Point (˚C/mmHg) 52
- Density (g/mL) 0.709
- Flash Point (˚C) -26 °C
- Refractive Index @ 20˚C 1.3880
Application
Review of synthetic utility.1
Reacts w/ aryl aldehydes to form diethynylmethane derivatives.10
Useful in the preparation of unsymmetrical diarylacetylenes.11
Ethynylates aromatic compounds.2
Precursor to trimethylsilylethynyl copper reagent.3
Lithiated derivative (n-BuLi treatment) reacts with halotriazines to produce monomers.4
Employed in ortho ethenylation of phenols.5
Undergoes Diels-Alder reactions with butadienes.6
Converts imines to propargyl amines.7
Forms propargylic amines from aldehydes and amines in aqueous system.8
Regioselectively forms either regioisomeric enyne upon addition to propargyl amines depending on catalyst employed.9
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 569-580.
10. Girard, D. et al. Tetrahedron Lett. 2007, 48, 6022.
11. Mio, M. et al. Org. Lett. 2002, 4, 3199.
2. Austin, W. et al. J. Org. Chem. 1981, 46, 2280.
3. Sakata, H. et al. Tetrahedron Lett. 1987, 28, 5719.
4. Kouveetakis, J. et al. Chem. Mater. 1994, 6, 636.
5. Kobayashi, K.; Yamaguchi, M. Org. Lett. 2001, 3, 241.
6. Paik, S.-J. et al. Org. Lett. 1999, 1, 2045.
7. Fischer, C.; Carreira, E. M. Org. Lett. 2001, 3, 4319.
8. Wei, C.; Li, C.-J. J. Am. Chem. Soc. 2003, 125, 9584.
9. Matsuyama, N. et al. J. Org. Chem. 2009, 74, 3576.
Safety
Alkynylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
Ethynyltrimethylsilane; Trimethylsilylacetylene
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products
Articles
In amongst the considerable chemistry of acetylenes there lies some unique chemistry of alkynylsilanes (silylacetylenes) some of which is reviewed herein. This unique character is exemplified not only in the silyl protection of the terminal C–H of acetylenes, but also in the ability of the silyl group to be converted into other functionalities after reaction of the alkynylsilane and to its ability to dictate and improve the regioselectivity of reactions at the triple bond. This, when combined with the possible subsequent transformations of the silyl group, makes their chemistry highly versatile and useful.