Additional Properties
- HMIS 3-3-1-X
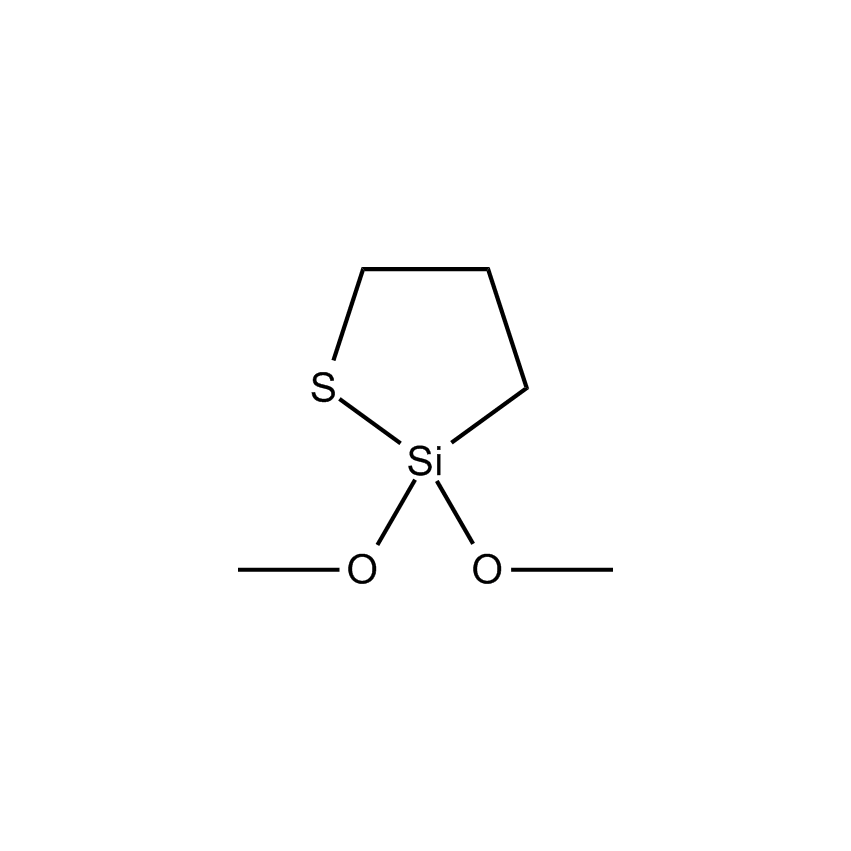
- Molecular Formula C5H12O2SSi
- Molecular Weight (g/mol) 164.29
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 57-58/7
- Density (g/mL) 1.094
Safety
2,2-Dimethoxy-1-thia-2-silacyclopentane
Applied Technology
Silicon Chemistry, Applied Technology
How cyclic azasilanes and cyclic thiasilanes, a new class of silane coupling agents, modify nanoparticles and create nano-scale features.
Silicon Chemistry, Applied Technology
Applied Technology
This overview provides an introduction and comparison of emerging processing technologies that represent the best contenders to satisfy future demands for ultrathin film applications.
Applied Technology
Silicon Chemistry, Applied Technology
Thiasilacyclopentane (TSCP) and azasilacyclopentane (ASCP) heteroatom cyclics reagents can be extended to “simultaneous doubleclicking” when both inorganic and organic substrates are present at the onset of the reaction. The simultaneous double-click depends on a first ring-opening click with an inorganic substrate that is complete in ~1 s at 30 °C and results in the reveal of a cryptic mercaptan or secondary amine group, which can then participate in a second click with an organic substrate.
Applied Technology