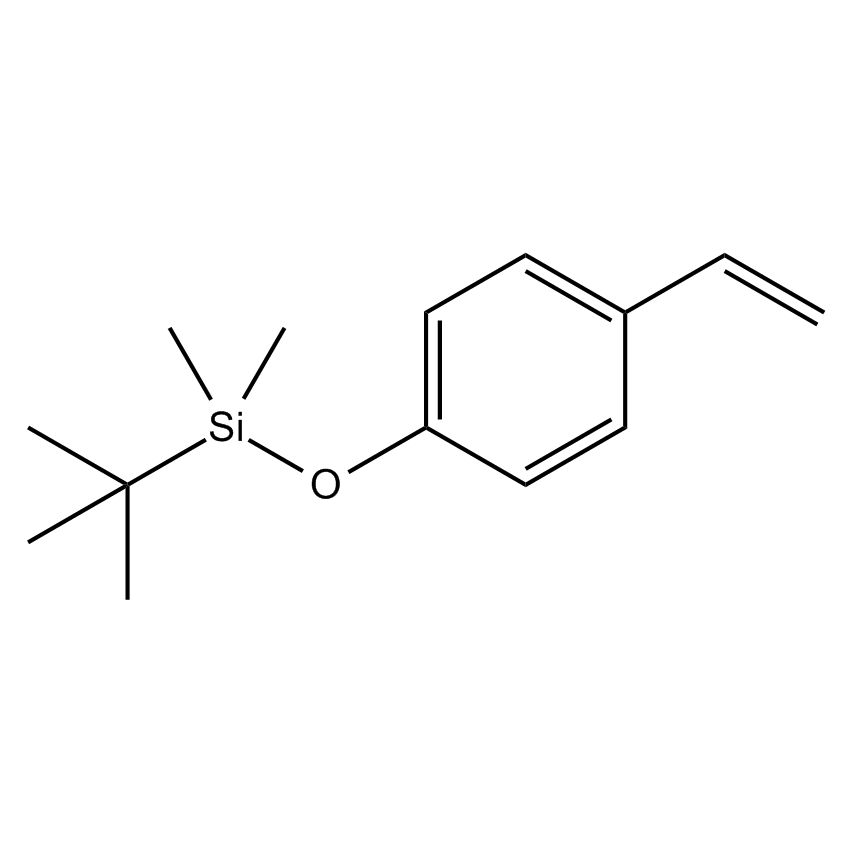
Additional Properties
- HMIS 2-2-0-X
- Molecular Formula C14H22OSi
- Molecular Weight (g/mol) 234.41
- Purity (%) 97%
- TSCA No
- Boiling Point (˚C/mmHg) 80/0.075
- Density (g/mL) 0.922
- Refractive Index @ 20˚C 1.5091
Application
Undergoes radical1,2 and anionic polymerization.3,4,5
Reference
1. Packirisamy, S. et al. J. Polym. Sci., Part A: Polym. Chem. 1989, 27, 2811.
2. Xu, Y. et al. Polymer 1991, 32, 3103.
3. Hirao, A. et al. Makromol. Chem., Rapid Commun. 1982, 3, 941.
4. Hirao, A. et al. Makromol. Chem. 1985, 186, 1157.
5. Ito, H. et al. Polym. Mater. Sci. Eng. Preprints 1993, 68, 12.
Safety
Alkenylsilane Cross-Coupling Agent
The cross-coupling reaction is a highly useful methodology for the formation of carbon-carbon bonds. It involves two reagents, with one typically being a suitable organometallic reagent - the nucleophile - and the other a suitable organic substrate, normally an unsaturated halide, tosylate or similar - the electrophile.
p-(t-Butyldimethylsiloxy)styrene; p-Vinyl-t-Butyldimethylbenzene
Silicon Chemistry, Articles
Hatanaka and Hiyama first reported the palladium-catalyzed, fluoride-promoted reaction of aryl, alkenyl, allyl, and ethynyltrimethylsilanes with aryl, vinil and allyl halides to form the respective cross-coupled products