Additional Properties
- Einecs Number 236-871-0
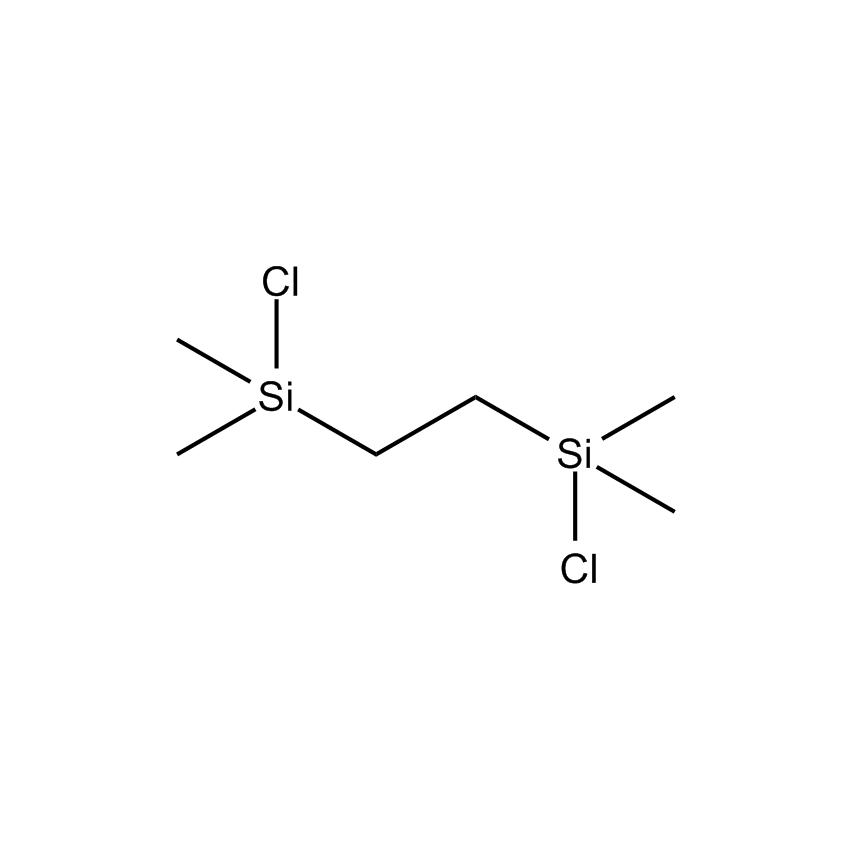
- Synonyms TETRAMETHYLDICHLORODISILETHYLENE
- HMIS 3-2-1-X
- Molecular Formula C6H16Cl2Si2
- Molecular Weight (g/mol) 215.27
- Purity (%) 97%
- TSCA Yes
- Boiling Point (˚C/mmHg) 198-199
- Flash Point (˚C) 40 °C
- Melting Point (˚C) 36-9°
Application
Review of synthetic utility.1
Reagent for protection of primary amines, including amino acids.2
Reference
1. Handbook of Reagents for Organic Synthesis, Reagents for Silicon-Mediated Organic Synthesis, Fuchs, P. L. Ed., John Wiley and Sons, Ltd., 2011, p. 48-50.
2. Djuric, S. et al. Tetrahedron Lett. 1981, 22, 1787.
Safety
Alkyl Silane - Dipodal Surface Bonding
Aliphatic, fluorinated aliphatic or substituted aromatic hydrocarbon substituents are the hydrophobic entities which enable silanes to induce surface hydrophobicity. The organic substitution of the silane must be non-polar. The hydrophobic effect of the organic substitution can be related to the free energy of transfer of hydrocarbon molecules from an aqueous phase to a homogeneous hydrocarbon phase. A successful hydrophobic coating must eliminate or mitigate hydrogen bonding and shield polar surfaces from interaction with water by creating a non-polar interphase. Although silane and silicone derived coatings are in general the most hydrophobic, they maintain a high degree of permeability to water vapor. This allows coatings to breathe and reduce deterioration at the coating interface associated with entrapped water. Since ions are not transported through non-polar silane and silicone coatings, they offer protection to composite structures ranging from pigmented coatings to rebar reinforced concrete. A selection guide for hydrophobic silanes can be found on pages 22-31 of the Hydrophobicity, Hydrophilicity and Silane Surface Modification brochure.
Bridging Silicon-Based Blocking Agent
Used as a protecting group for reactive hydrogens in alcohols, amines, thiols, and carboxylic acids. Organosilanes are hydrogen-like, can be introduced in high yield, and can be removed under selective conditions. They are stable over a wide range of reaction conditions and can be removed in the presence of other functional groups, including other protecting groups. The tolerance of silylated alcohols to chemical transformations summary is presented in Table 1 of the Silicon-Based Blocking Agents brochure.
Dipodal Silane
Dipodal silanes are a series of adhesion promoters that have intrinsic hydrolytic stabilities up to ~10,000 times greater than conventional silanes and are used in applications such as plastic optics, multilayer printed circuit boards and as adhesive primers for ferrous and nonferrous metals. They have the ability to form up to six bonds to a substrate compared to conventional silanes with the ability to form only three bonds to a substrate. Many conventional coupling agents are frequently used in combination with 10-40% of a non-functional dipodal silane, where the conventional coupling agent provides the appropriate functionality for the application, and the non-functional dipodal silane provides increased durability. Also known as bis-silanes additives enhance hydrolytic stability, which impacts on increased product shelf life, ensures better substrate bonding and also leads to improved mechanical properties in coatings as well as composite applications.
Bis(dimethylchlorosilyl)ethane; Tetramethyldichlorodisilethylene; Ethylenebis[chlorodimethylsilane]; STABASE-Cl