Additional Properties
- HMIS 3-1-1-X
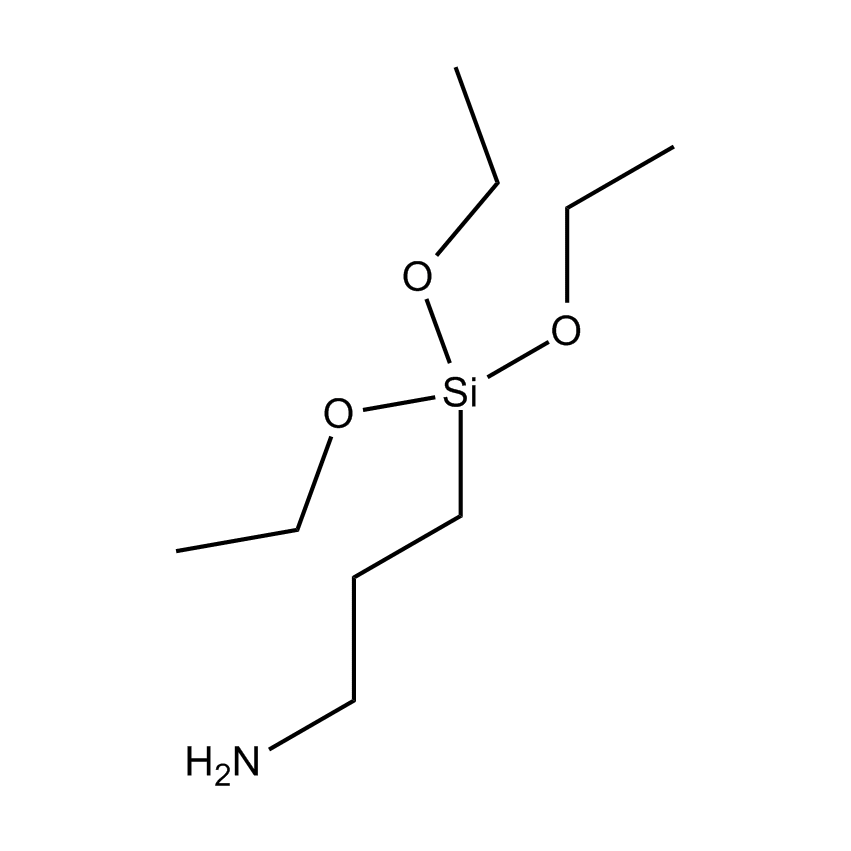
- Molecular Formula C9H23NO3Si
- Molecular Weight (g/mol) 221.37
- TSCA Yes
- Boiling Point (˚C/mmHg) 122-123/30
- Density (g/mL) 0.948
- Flash Point (˚C) 91 °C
- Viscosity at 25 ˚C (cSt) 2.0
Safety
SIVATE E610 (Enhanced AMEO)
Enhanced Amine Functional Trialkoxy Silane
Silane coupling agents have the ability to form a durable bond between organic and inorganic materials to generate desired heterogeneous environments or to incorporate the bulk properties of different phases into a uniform composite structure. The general formula has two classes of functionality. The hydrolyzable group forms stable condensation products with siliceous surfaces and other oxides such as those of aluminum, zirconium, tin, titanium, and nickel. The organofunctional group alters the wetting or adhesion characteristics of the substrate, utilizes the substrate to catalyze chemical transformations at the heterogeneous interface, orders the interfacial region, or modifies its partition characteristics, and significantly effects the covalent bond between organic and inorganic materials.
Archived, Publications & Technical Reports
Applied Technology