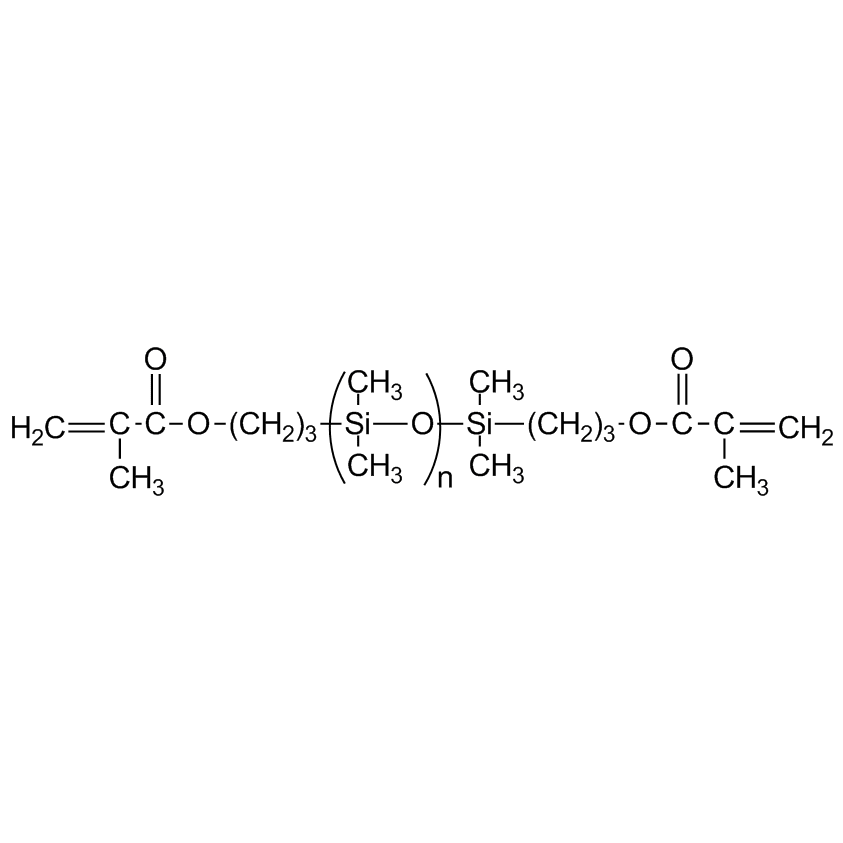Safety
- HMIS 2-1-0-X
- Molecular Weight (g/mol) 900-1,200
- TSCA No
- Boiling Point (˚C/mmHg) NA
- Density (g/mL) 0.98
- Refractive Index @ 20˚C 1.422
- Viscosity at 25 ˚C (cSt) 8-14
DMS-R11: Methacryloxypropyl Terminated PDMS, 8-14 cSt (Telechelic Functional Fluid)
Methacrylate and Acrylate functional siloxanes undergo the same reactions generally associated with methacrylates and acrylates, the most conspicuous being radical induced polymerization. Unlike vinylsiloxanes which are sluggish compared to their organic counterparts, methacrylate and acrylate siloxanes have similar reactivity to their organic counterparts. The principal applications of methacrylate functional siloxanes are as modifiers to organic systems. Upon radical induced polymerization, methacryloxypropyl terminated siloxanes by themselves only increase in viscosity. Copolymers with greater than 5 mole % methacrylate substitution crosslink to give non-flowable resins. Acrylate functional siloxanes cure greater than ten times as fast methacrylate functional siloxanes on exposure to UV in the presence of a photoinitiator such as ethylbenzoin. Oxygen is an inhibitor for methacrylate polymerization in general. The high oxygen permeability of siloxanes usually makes it necessary to blanket these materials with nitrogen or argon in order to obtain reasonable cures.
DMS-R11 Properties