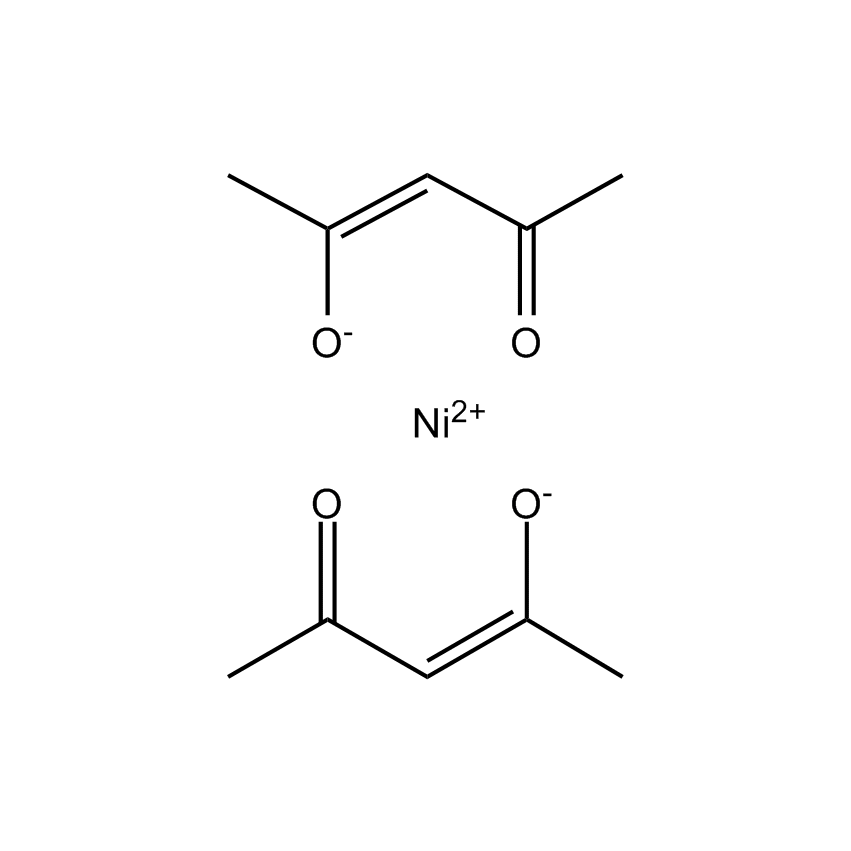
Additional Properties
- Einecs Number 221-875-7
- HMIS 3-1-0-X
- Molecular Formula C10H14NiO4
- Molecular Weight (g/mol) 256.91
- TSCA Yes
- Boiling Point (˚C/mmHg) NA
- Density (g/mL) 1.455
- Melting Point (˚C) 208° dec
- Refractive Index @ 20˚C 1.57-1.64
Application
Catalyzes conjugate addition of alkynyl aluminums to enones.1
Catalyzes coupling of Grignard reagents to give biaryls.2
Catalyzes Grignard additions to silyl enol ethers forming alkenes.3
Thermochromic effect in non-coordinating solvents at 200°C.4
UV stabilizer for polyphenylene sulfide.5
Catalyzes coupling of dialkylzincs with alkyl iodides.6
Intermediate for Ni nanoparticles by reduction with NaBH4.7
Forms Ni3C nanocrystals by thermolysis in oleylamine.8
Reference
1. Hansen, R. T. et al. J. Am. Chem. Soc. 1978, 100, 2244.
2. Clough, R. L. J. Org. Chem. 1976, 41, 2252.
3. Hayashi, T. et al. Tetrahedron Lett. 1980, 3915.
4. Cotton, F. et al. J. Am. Chem. Soc. 1961, 83, 2818.
5. Stahlke, K. Chem. Abstr. 114, 144761u; Eur. Patent Application 326,888, 1989.
6. Giovannini, G. R. et al. J. Org. Chem. 1999, 64, 3544.
7. Green, M. et al. J. Chem. Soc., Chem. Commun. 2001, 1912.
8. Goto, Y. et al. Chem. Mater. 2008, 20, 4156.
Safety
ALD Material
Atomic layer deposition (ALD) is a chemically self-limiting deposition technique that is based on the sequential use of a gaseous chemical process. A thin film (as fine as -0.1 Å per cycle) results from repeating the deposition sequence as many times as needed to reach a certain thickness. The major characteristic of the films is the resulting conformality and the controlled deposition manner. Precursor selection is key in ALD processes, namely finding molecules which will have enough reactivity to produce the desired films yet are stable enough to be handled and safely delivered to the reaction chamber.
Nickel(II) 2,4-pentanedionate; Acetylacetonate: nickel acac; Bis(pentane-2,4-dionato-O,O')nickel; Nickel(II)bis(4-hydroxy-2-pentanolate)